In vitro developmental potential of macaque oocytes, derived from unstimulated ovaries, following maturation in the presence of glutathione ethyl ester
- PMID: 20729236
- PMCID: PMC2939760
- DOI: 10.1093/humrep/deq225
In vitro developmental potential of macaque oocytes, derived from unstimulated ovaries, following maturation in the presence of glutathione ethyl ester
Abstract
Background: The inadequacies of oocyte in vitro maturation (IVM) systems for both non-human primates and humans are evidenced by reduced fertilization and poor embryonic development, and may be partly explained by significantly lower glutathione (GSH) contents compared with in vivo matured (IVO) oocytes. As this influence has not been fully explored, this study investigated the effect of the GSH donor, glutathione ethyl ester (GSH-OEt), on the IVM and development of macaque oocytes as a model of human oocyte IVM.
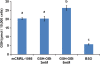
Methods: Macaque oocytes derived from unstimulated ovaries were cultured in mCMRL-1066 alone or supplemented with 3 or 5 mM GSH-OEt. In vitro matured oocytes were subjected to the GSH assay, fixed for the assessment of spindle morphology or prepared ICSI. Embryo development of zygotes cultured in mHECM-9 was assessed up to Day 9 post-ICSI. RESULTS Supplementation of the maturation medium with GSH-OEt significantly increased oocyte maturation and normal fertilization rates compared with control oocytes, but only 5 mM GSH-OEt significantly increased the oocyte and cumulus cell GSH content. Confocal microscopy revealed significant differences in the spindle morphology between IVO and control in vitro matured metaphase II oocytes. Oocytes matured with 5 mM GSH-OEt exhibited spindle area and spindle pole width similar to that seen in the IVO oocyte. While no significant differences were observed in blastocyst rates, addition of 3 mM GSH-OEt during IVM significantly increased the proportion of embryos developing to the 5-8 cell stage while 5 mM GSH-OEt significantly increased the proportion of morula-stage embryos compared with controls.
Conclusions: Supplementation of the IVM medium with GSH-OEt promotes better maturation and normal fertilization of macaque oocytes compared with non-supplemented medium. However, further improvement of the primate oocyte IVM culture system is required to support better blastocyst development of oocytes derived from unstimulated ovaries.
Figures



Similar articles
-
Primate model of metaphase I oocyte in vitro maturation and the effects of a novel glutathione donor on maturation, fertilization, and blastocyst development.Fertil Steril. 2011 Mar 15;95(4):1235-40. doi: 10.1016/j.fertnstert.2010.06.029. Epub 2010 Jul 29. Fertil Steril. 2011. PMID: 20667536 Free PMC article.
-
Improved cryotolerance and developmental potential of in vitro and in vivo matured mouse oocytes by supplementing with a glutathione donor prior to vitrification.Mol Hum Reprod. 2016 Dec;22(12):867-881. doi: 10.1093/molehr/gaw059. Epub 2016 Sep 7. Mol Hum Reprod. 2016. PMID: 27604460
-
Bovine in vitro oocyte maturation as a model for manipulation of the gamma-glutamyl cycle and intraoocyte glutathione.Reprod Fertil Dev. 2008;20(5):579-88. doi: 10.1071/rd08041. Reprod Fertil Dev. 2008. PMID: 18577355
-
The influence of reduced glutathione in fertilization medium on the fertility of in vitro-matured C57BL/6 mouse oocytes.Theriogenology. 2013 Sep 15;80(5):421-6. doi: 10.1016/j.theriogenology.2013.07.002. Epub 2013 Aug 2. Theriogenology. 2013. PMID: 23916252 Review.
-
Contribution of human oocyte architecture to success of in vitro maturation technology.Iran J Reprod Med. 2013 Jan;11(1):1-10. Iran J Reprod Med. 2013. PMID: 24639686 Free PMC article. Review.
Cited by
-
Extensive effects of in vitro oocyte maturation on rhesus monkey cumulus cell transcriptome.Am J Physiol Endocrinol Metab. 2011 Jul;301(1):E196-209. doi: 10.1152/ajpendo.00686.2010. Epub 2011 Apr 12. Am J Physiol Endocrinol Metab. 2011. PMID: 21487073 Free PMC article.
-
In vitro matured oocytes are more susceptible than in vivo matured oocytes to mock ICSI induced functional and genetic changes.PLoS One. 2015 Mar 18;10(3):e0119735. doi: 10.1371/journal.pone.0119735. eCollection 2015. PLoS One. 2015. PMID: 25786120 Free PMC article.
-
Decidual Stromal Cell Ferroptosis Associated with Abnormal Iron Metabolism Is Implicated in the Pathogenesis of Recurrent Pregnancy Loss.Int J Mol Sci. 2023 Apr 25;24(9):7836. doi: 10.3390/ijms24097836. Int J Mol Sci. 2023. PMID: 37175543 Free PMC article.
-
Ceratonia siliqua (Carob) extract improved in vitro development of vitrified-warmed mouse germinal vesicle oocytes: assessment of possible mechanism.Cell Tissue Bank. 2021 Mar;22(1):137-144. doi: 10.1007/s10561-020-09873-w. Epub 2020 Oct 14. Cell Tissue Bank. 2021. PMID: 33052521
-
Induced pluripotent stem cells from highly endangered species.Nat Methods. 2011 Sep 4;8(10):829-31. doi: 10.1038/nmeth.1706. Nat Methods. 2011. PMID: 21892153
References
-
- Abeydeera LR, Wang W-H, Cantley TC, Prather RS, Day BN. Glutathione content and embryo development after in vitro fertilization of pig oocytes matured in the presence of a thiol compound and various concentrations of cysteine. Zygote. 1999;7:203–210. - PubMed
-
- Agarwal A, Gupta S, Sekhon L, Shah R. Redox considerations in female reproductive function and assisted reproduction: from molecular mechanisms to health implications. Antioxid Redox Signal. 2008;10:1375–1403. - PubMed
-
- Albertini DF. Cytoplasmic microtubular dynamics and chromatin organization during mammalian oogenesis and oocyte maturation. Mutat Res. 1992;296:57–68. - PubMed
-
- Allansson L, Khatibi S, Olsson T, Hansson E. Acute ethanol exposure induces [Ca2+]i transients, cell swelling and transformation of actin cytoskeleton in astroglial primary cultures. J Neurochem. 2001;76:472–479. - PubMed
-
- Anderson ME, Powrie F, Puri RN, Meister A. Glutathione monoethyl ester: preparation, uptake by tissues, and conversion to glutathione. Arch Biochem Biophys. 1985;239:538–548. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

