Deficiency of insulin-like growth factor 1 reduces vulnerability to chronic alcohol intake-induced cardiomyocyte mechanical dysfunction: role of AMPK
- PMID: 20731752
- PMCID: PMC3017727
- DOI: 10.1111/j.1582-4934.2010.01158.x
Deficiency of insulin-like growth factor 1 reduces vulnerability to chronic alcohol intake-induced cardiomyocyte mechanical dysfunction: role of AMPK
Abstract
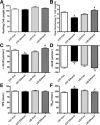
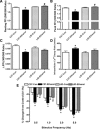
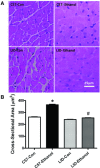
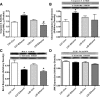
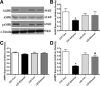
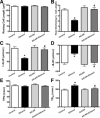
Circulating insulin-like growth factor I (IGF-1) levels are closely associated with cardiac performance although the role of IGF-1 in alcoholic cardiac dysfunction is unknown. This study was designed to evaluate the impact of severe liver IGF-1 deficiency (LID) on chronic alcohol-induced cardiomyocyte contractile and intracellular Ca(2+) dysfunction. Adult male C57 and LID mice were placed on a 4% alcohol diet for 15 weeks. Cardiomyocyte contractile and intracellular Ca(2+) properties were evaluated including peak shortening (PS), maximal velocity of shortening/relengthening (±dL/dt), time-to-relengthening (TR(90) ), change in fura-fluorescence intensity (ΔFFI) and intracellular Ca(2+) decay. Levels of apoptotic regulators caspase-3, Bcl-2 and c-Jun NH2-terminal kinase (JNK), the ethanol metabolizing enzyme mitochondrial aldehyde dehydrogenase (ALDH2), as well as the cellular fuel gauge AMP-activated protein kinase (AMPK) were evaluated. Chronic alcohol intake enlarged myocyte cross-sectional area, reduced PS, ± dL/dt and ΔFFI as well as prolonged TR(90) and intracellular Ca(2+) decay, the effect of which was greatly attenuated by IGF-1 deficiency. The beneficial effect of LID against alcoholic cardiac mechanical defect was ablated by IGF-1 replenishment. Alcohol intake increased caspase-3 activity/expression although it down-regulated Bcl-2, ALDH2 and pAMPK without affecting JNK and AMPK. IGF-1 deficiency attenuated alcoholism-induced responses in all these proteins with the exception of Bcl-2. In addition, the AMPK agonist 5-aminoimidazole-4-carboxamide-1-β-D-ribofuranoside abrogated short-term ethanol incubation-elicited cardiac mechanical dysfunction. Taken together, these data suggested that IGF-1 deficiency may reduce the sensitivity to ethanol-induced myocardial mechanical dysfunction. Our data further depicted a likely role of Caspase-3, ALDH2 and AMPK activation in IGF-1 deficiency induced 'desensitization' of alcoholic cardiomyopathy.
© 2011 The Authors Journal compilation © 2011 Foundation for Cellular and Molecular Medicine/Blackwell Publishing Ltd.
Figures

Similar articles
-
Insulin-like growth factor I (IGF-1) deficiency ameliorates sex difference in cardiac contractile function and intracellular Ca(2+) homeostasis.Toxicol Lett. 2011 Oct 10;206(2):130-8. doi: 10.1016/j.toxlet.2011.07.001. Epub 2011 Jul 7. Toxicol Lett. 2011. PMID: 21763763 Free PMC article.
-
Deficiency of insulin-like growth factor 1 reduces sensitivity to aging-associated cardiomyocyte dysfunction.Rejuvenation Res. 2008 Aug;11(4):725-33. doi: 10.1089/rej.2008.0717. Rejuvenation Res. 2008. PMID: 18729805 Free PMC article.
-
Cardiac-specific overexpression of insulin-like growth factor I (IGF-1) rescues lipopolysaccharide-induced cardiac dysfunction and activation of stress signaling in murine cardiomyocytes.Shock. 2009 Jul;32(1):100-7. doi: 10.1097/SHK.0b013e31818ec609. Shock. 2009. PMID: 18948844 Free PMC article.
-
17-β estradiol attenuates ovariectomy-induced changes in cardiomyocyte contractile function via activation of AMP-activated protein kinase.Toxicol Lett. 2015 Jan 5;232(1):253-62. doi: 10.1016/j.toxlet.2014.11.012. Epub 2014 Nov 13. Toxicol Lett. 2015. PMID: 25448287 Free PMC article.
-
Cardiac overexpression of metallothionein attenuates chronic alcohol intake-induced cardiomyocyte contractile dysfunction.Cardiovasc Toxicol. 2006;6(3-4):173-82. doi: 10.1385/ct:6:3:173. Cardiovasc Toxicol. 2006. PMID: 17347528
Cited by
-
Mechanisms Underlying Muscle Protein Imbalance Induced by Alcohol.Annu Rev Nutr. 2018 Aug 21;38:197-217. doi: 10.1146/annurev-nutr-071816-064642. Annu Rev Nutr. 2018. PMID: 30130465 Free PMC article.
-
ALDH2 in alcoholic heart diseases: molecular mechanism and clinical implications.Pharmacol Ther. 2011 Oct;132(1):86-95. doi: 10.1016/j.pharmthera.2011.05.008. Epub 2011 Jun 12. Pharmacol Ther. 2011. PMID: 21664374 Free PMC article. Review.
-
Aldehyde dedydrogenase-2 plays a beneficial role in ameliorating chronic alcohol-induced hepatic steatosis and inflammation through regulation of autophagy.J Hepatol. 2015 Mar;62(3):647-56. doi: 10.1016/j.jhep.2014.10.009. Epub 2014 Oct 20. J Hepatol. 2015. Retraction in: J Hepatol. 2023 Oct;79(4):1072. doi: 10.1016/j.jhep.2023.08.011. PMID: 25457208 Free PMC article. Retracted.
-
Targeting the energy guardian AMPK: another avenue for treating cardiomyopathy?Cell Mol Life Sci. 2017 Apr;74(8):1413-1429. doi: 10.1007/s00018-016-2407-7. Epub 2016 Nov 4. Cell Mol Life Sci. 2017. PMID: 27815596 Free PMC article. Review.
-
Insulin-like growth factor I (IGF-1) deficiency ameliorates sex difference in cardiac contractile function and intracellular Ca(2+) homeostasis.Toxicol Lett. 2011 Oct 10;206(2):130-8. doi: 10.1016/j.toxlet.2011.07.001. Epub 2011 Jul 7. Toxicol Lett. 2011. PMID: 21763763 Free PMC article.
References
-
- Ren J, Samson WK, Sowers JR. Insulin-like growth factor I as a cardiac hormone: physiological and pathophysiological implications in heart disease. J Mol Cell Cardiol. 1999;31:2049–61. - PubMed
-
- Gola M, Bonadonna S, Doga M, et al. Cardiovascular risk in aging and obesity: is there a role for GH. J Endocrinol Invest. 2005;28:759–67. - PubMed
-
- Yakar S, Sun H, Zhao H, et al. Metabolic effects of IGF-I deficiency: lessons from mouse models. Pediatr Endocrinol Rev. 2005;3:11–9. - PubMed
-
- Climent V, Marin F, Pico A, et al. Growth hormone therapy, is it always good for the heart. Eur Heart J. 2004;25:183. - PubMed
-
- Li Q, Yang X, Sreejayan N, et al. Insulin-like growth factor I deficiency prolongs survival and antagonizes paraquat-induced cardiomyocyte dysfunction: role of oxidative stress. Rejuvenation Res. 2007;10:501–12. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

