Effects of endoplasmic reticulum stress on group VIA phospholipase A2 in beta cells include tyrosine phosphorylation and increased association with calnexin
- PMID: 20732873
- PMCID: PMC2962484
- DOI: 10.1074/jbc.M110.153197
Effects of endoplasmic reticulum stress on group VIA phospholipase A2 in beta cells include tyrosine phosphorylation and increased association with calnexin
Abstract
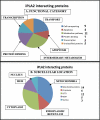
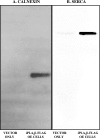
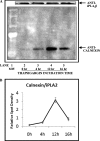
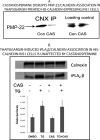
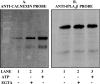
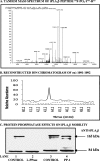
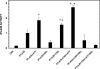
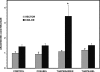
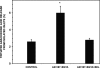
The Group VIA phospholipase A(2) (iPLA(2)β) hydrolyzes glycerophospholipids at the sn-2-position to yield a free fatty acid and a 2-lysophospholipid, and iPLA(2)β has been reported to participate in apoptosis, phospholipid remodeling, insulin secretion, transcriptional regulation, and other processes. Induction of endoplasmic reticulum (ER) stress in β-cells and vascular myocytes with SERCA inhibitors activates iPLA(2)β, resulting in hydrolysis of arachidonic acid from membrane phospholipids, by a mechanism that is not well understood. Regulatory proteins interact with iPLA(2)β, including the Ca(2+)/calmodulin-dependent protein kinase IIβ, and we have characterized the iPLA(2)β interactome further using affinity capture and LC/electrospray ionization/MS/MS. An iPLA(2)β-FLAG fusion protein was expressed in an INS-1 insulinoma cell line and then adsorbed to an anti-FLAG matrix after cell lysis. iPLA(2)β and any associated proteins were then displaced with FLAG peptide and analyzed by SDS-PAGE. Gel sections were digested with trypsin, and the resultant peptide mixtures were analyzed by LC/MS/MS with database searching. This identified 37 proteins that associate with iPLA(2)β, and nearly half of them reside in ER or mitochondria. They include the ER chaperone calnexin, whose association with iPLA(2)β increases upon induction of ER stress. Phosphorylation of iPLA(2)β at Tyr(616) also occurs upon induction of ER stress, and the phosphoprotein associates with calnexin. The activity of iPLA(2)β in vitro increases upon co-incubation with calnexin, and overexpression of calnexin in INS-1 cells results in augmentation of ER stress-induced, iPLA(2)β-catalyzed hydrolysis of arachidonic acid from membrane phospholipids, reflecting the functional significance of the interaction. Similar results were obtained with mouse pancreatic islets.
Figures



References
-
- Tang J., Kriz R. W., Wolfman N., Shaffer M., Seehra J., Jones S. S. (1997) J. Biol. Chem. 272, 8567–8575 - PubMed
-
- Balboa M. A., Balsinde J., Jones S. S., Dennis E. A. (1997) J. Biol. Chem. 272, 8576–8580 - PubMed
-
- Ma Z., Ramanadham S., Kempe K., Chi X. S., Ladenson J., Turk J. (1997) J. Biol. Chem. 272, 11118–11127 - PubMed
-
- Yang H. C., Mosior M., Ni B., Dennis E. A. (1999) J. Neurochem. 73, 1278–1287 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

