α6ß2* and α4ß2* nicotinic receptors both regulate dopamine signaling with increased nigrostriatal damage: relevance to Parkinson's disease
- PMID: 20732972
- PMCID: PMC2981368
- DOI: 10.1124/mol.110.067561
α6ß2* and α4ß2* nicotinic receptors both regulate dopamine signaling with increased nigrostriatal damage: relevance to Parkinson's disease
Abstract
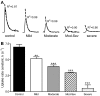
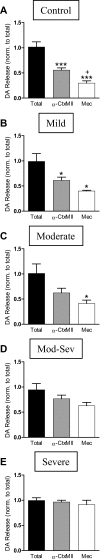
Nicotinic receptors (nAChRs) are important modulators of dopaminergic transmission in striatum, a region critical to Parkinson's disease. The nAChRs mainly involved are the α6β2* and α4β2* subtypes. Lesion studies show that the α6β2* receptor is decreased to a much greater extent with nigrostriatal damage than the α4β2* subtype raising the question whether this latter nAChR population is more important with increased nigrostriatal damage. To address this, we investigated the effect of varying nigrostriatal damage on α6β2* and α4β2* receptor-modulated dopamine signaling using cyclic voltammetry. This approach offers the advantage that changes in dopamine release can be observed under different neuronal firing conditions. Total single-pulse-evoked dopamine release decreased in direct proportion to declines in the dopamine transporter and dopamine uptake. We next used α-conotoxinMII and mecamylamine to understand the role of the α4β2* and α6β2* subtypes in release. Single-pulse-stimulated α6β2* and α4β2* receptor dopamine release decreased to a similar extent with increasing nigrostriatal damage, indicating that both subtypes contribute to the control of dopaminergic transmission with lesioning. Total burst-stimulated dopamine release also decreased proportionately with nigrostriatal damage. However, the role of the α4β2* and α6β2* nAChRs varied with different degrees of lesioning, suggesting that the two subtypes play a unique function with burst firing, with a somewhat more prominent and possibly more selective role for the α6β2* subtype. These data have important therapeutic implications because they suggest that drugs directed to both α4β2* and α6β2* nAChRs may be useful in the treatment of neurological disorders such as Parkinson's disease.
Figures
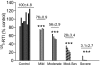



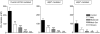
Similar articles
-
Nigrostriatal damage preferentially decreases a subpopulation of alpha6beta2* nAChRs in mouse, monkey, and Parkinson's disease striatum.Mol Pharmacol. 2007 Jul;72(1):52-61. doi: 10.1124/mol.107.035998. Epub 2007 Apr 4. Mol Pharmacol. 2007. PMID: 17409284
-
Decrease in alpha3*/alpha6* nicotinic receptors but not nicotine-evoked dopamine release in monkey brain after nigrostriatal damage.Mol Pharmacol. 2005 Sep;68(3):737-46. doi: 10.1124/mol.105.012773. Epub 2005 Jun 2. Mol Pharmacol. 2005. PMID: 15933214
-
Nicotine partially protects against paraquat-induced nigrostriatal damage in mice; link to alpha6beta2* nAChRs.J Neurochem. 2007 Jan;100(1):180-90. doi: 10.1111/j.1471-4159.2006.04177.x. J Neurochem. 2007. PMID: 17227438
-
Multiple roles for nicotine in Parkinson's disease.Biochem Pharmacol. 2009 Oct 1;78(7):677-85. doi: 10.1016/j.bcp.2009.05.003. Epub 2009 May 9. Biochem Pharmacol. 2009. PMID: 19433069 Free PMC article. Review.
-
Nicotinic receptors as CNS targets for Parkinson's disease.Biochem Pharmacol. 2007 Oct 15;74(8):1224-34. doi: 10.1016/j.bcp.2007.06.015. Epub 2007 Jun 17. Biochem Pharmacol. 2007. PMID: 17631864 Free PMC article. Review.
Cited by
-
Nicotinic Receptor Intervention in Parkinson's Disease: Future Directions.Clin Pharmacol Transl Med. 2017;1(1):14-19. Epub 2017 Mar 6. Clin Pharmacol Transl Med. 2017. PMID: 29863173 Free PMC article.
-
The striatal cholinergic system in L-dopa-induced dyskinesias.J Neural Transm (Vienna). 2018 Aug;125(8):1251-1262. doi: 10.1007/s00702-018-1845-9. Epub 2018 Feb 28. J Neural Transm (Vienna). 2018. PMID: 29492663 Free PMC article. Review.
-
Loss of Homeostasis in the Direct Pathway in a Mouse Model of Asymptomatic Parkinson's Disease.J Neurosci. 2016 May 25;36(21):5686-98. doi: 10.1523/JNEUROSCI.0492-15.2016. J Neurosci. 2016. PMID: 27225760 Free PMC article.
-
Examining the complex regulation and drug-induced plasticity of dopamine release and uptake using voltammetry in brain slices.ACS Chem Neurosci. 2013 May 15;4(5):693-703. doi: 10.1021/cn400026v. Epub 2013 May 6. ACS Chem Neurosci. 2013. PMID: 23581570 Free PMC article. Review.
-
Varenicline enhances dopamine release facilitation more than nicotine after long-term nicotine treatment and withdrawal.Pharmacol Res Perspect. 2015 Feb;3(1):e00105. doi: 10.1002/prp2.105. Epub 2014 Dec 16. Pharmacol Res Perspect. 2015. PMID: 25692023 Free PMC article.
References
-
- Artymyshyn R, Smith A, Wolfe BB. (1990) The use of 3H standards in 125I autoradiography. J Neurosci Methods 32:185–192 - PubMed
-
- Bergstrom BP, Schertz KE, Weirick T, Nafziger B, Takacs SA, Lopes KO, Massa KJ, Walker QD, Garris PA. (2001) Partial, graded losses of dopamine terminals in the rat caudate-putamen: an animal model for the study of compensatory adaptation in preclinical parkinsonism. J Neurosci Methods 106:15–28 - PubMed
-
- Bordia T, Campos C, Huang L, Quik M. (2008) Continuous and intermittent nicotine treatment reduces l-3,4-dihydroxyphenylalanine (l-DOPA)-induced dyskinesias in a rat model of Parkinson's disease. J Pharmacol Exp Ther 327:239–247 - PubMed
-
- Bordia T, Grady SR, McIntosh JM, Quik M. (2007) Nigrostriatal damage preferentially decreases a subpopulation of alpha6beta2* nAChRs in mouse, monkey, and Parkinson's disease striatum. Mol Pharmacol 72:52–61 - PubMed
-
- Cenci MA, Lundblad M. (2007) Ratings of L-DOPA-induced dyskinesia in the unilateral 6-OHDA lesion model of Parkinson's disease in rats and mice. Curr Protoc Neurosci Chapter 9:Unit 9.25 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

