The cystine/glutamate antiporter regulates dendritic cell differentiation and antigen presentation
- PMID: 20733204
- PMCID: PMC3100200
- DOI: 10.4049/jimmunol.1001199
The cystine/glutamate antiporter regulates dendritic cell differentiation and antigen presentation
Abstract
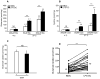
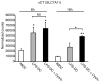
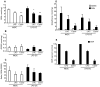
The major cellular antioxidant glutathione is depleted during HIV infection and in obesity. Although the consequence of glutathione depletion on immune function is starting to emerge, it is currently not known whether glutathione dysregulation influences the differentiation and maturation of dendritic cells (DCs). Moreover, the effect of glutathione depletion on DC effector functions, such as Ag presentation, is poorly understood. Glutathione synthesis depends on the cystine/glutamate antiporter, which transports the rate-limiting precursor cystine into the cell in exchange for glutamate. In this paper, we present a detailed study of antiporter function in DCs and demonstrate a role for the antiporter in DC differentiation and cross-presentation. We show that the antiporter is the major mechanism for transport of cystine and glutamate and modulates the intracellular glutathione content and glutathione efflux from DCs. Blocking antiporter-dependent cystine transport decreases intracellular glutathione levels, and these effects correlate with reduced transcription of the functional subunit of the antiporter. We further demonstrate that blocking antiporter activity interferes with DC differentiation from monocyte precursors, but antiporter activity is not required for LPS-induced phenotypic maturation. Finally, we show that inhibiting antiporter uptake of cystine interferes with presentation of exogenous Ag to class II MHC-restricted T cells and blocks cross-presentation on MHC class I. We conclude that aberrant antiporter function disrupts glutathione homeostasis in DCs and may contribute to impaired immunity in the diseased host.
Figures
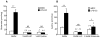
Similar articles
-
The cystine/glutamate antiporter regulates the functional expression of indoleamine 2,3-dioxygenase in human dendritic cells.Scand J Immunol. 2012 Oct;76(4):448-9. doi: 10.1111/j.1365-3083.2012.02743.x. Scand J Immunol. 2012. PMID: 22690871 Free PMC article. No abstract available.
-
The cystine/glutamate antiporter regulates indoleamine 2,3-dioxygenase protein levels and enzymatic activity in human dendritic cells.Am J Clin Exp Immunol. 2012 Nov 30;1(2):113-123. Am J Clin Exp Immunol. 2012. PMID: 23243629 Free PMC article.
-
Selection of an antibody library identifies a pathway to induce immunity by targeting CD36 on steady-state CD8 alpha+ dendritic cells.J Immunol. 2008 Mar 1;180(5):3201-9. doi: 10.4049/jimmunol.180.5.3201. J Immunol. 2008. PMID: 18292544
-
Mechanisms of oxidative glutamate toxicity: the glutamate/cystine antiporter system xc- as a neuroprotective drug target.CNS Neurol Disord Drug Targets. 2010 Jul;9(3):373-82. doi: 10.2174/187152710791292567. CNS Neurol Disord Drug Targets. 2010. PMID: 20053169 Review.
-
Thinking outside the cleft to understand synaptic activity: contribution of the cystine-glutamate antiporter (System xc-) to normal and pathological glutamatergic signaling.Pharmacol Rev. 2012 Jul;64(3):780-802. doi: 10.1124/pr.110.003889. Pharmacol Rev. 2012. PMID: 22759795 Free PMC article. Review.
Cited by
-
Aspartate Metabolism Facilitates IL-1β Production in Inflammatory Macrophages.Front Immunol. 2021 Oct 21;12:753092. doi: 10.3389/fimmu.2021.753092. eCollection 2021. Front Immunol. 2021. PMID: 34745126 Free PMC article.
-
Neurotransmitter signaling in the pathophysiology of microglia.Front Cell Neurosci. 2013 Apr 19;7:49. doi: 10.3389/fncel.2013.00049. eCollection 2013. Front Cell Neurosci. 2013. PMID: 23626522 Free PMC article.
-
Metabolic Regulation of Dendritic Cell Differentiation.Front Immunol. 2019 Mar 13;10:410. doi: 10.3389/fimmu.2019.00410. eCollection 2019. Front Immunol. 2019. PMID: 30930893 Free PMC article. Review.
-
Impact of the Type I Interferon Receptor on the Global Gene Expression Program During the Course of Dendritic Cell Maturation Induced by Polyinosinic Polycytidylic Acid.J Interferon Cytokine Res. 2016 Jun;36(6):382-400. doi: 10.1089/jir.2014.0150. Epub 2016 Apr 1. J Interferon Cytokine Res. 2016. PMID: 27035059 Free PMC article.
-
Dendritic cell immunometabolism - a potential therapeutic target for allergic diseases.Int J Med Sci. 2025 Jan 1;22(2):417-431. doi: 10.7150/ijms.105532. eCollection 2025. Int J Med Sci. 2025. PMID: 39781535 Free PMC article. Review.
References
-
- Meister A, Anderson ME. Glutathione. Annu Rev Biochem. 1983;52:711–760. - PubMed
-
- Reed DJ. Regulation of reductive processes by glutathione. Biochem Pharmacol. 1986;35:7–13. - PubMed
-
- Jones DP. Redox potential of GSH/GSSG couple: assay and biological significance. Methods Enzymol. 2002;348:93–112. - PubMed
-
- Klebanoff SJ. Oxygen metabolism and the toxic properties of phagocytes. Ann Intern Med. 1980;93:480–489. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

