Dual bronchodilatory and pulmonary anti-inflammatory activity of RO5024118, a novel agonist at vasoactive intestinal peptide VPAC2 receptors
- PMID: 20735404
- PMCID: PMC3000657
- DOI: 10.1111/j.1476-5381.2010.00975.x
Dual bronchodilatory and pulmonary anti-inflammatory activity of RO5024118, a novel agonist at vasoactive intestinal peptide VPAC2 receptors
Abstract
Background and purpose: Vasoactive intestinal peptide is expressed in the respiratory tract and induces its effects via its receptors, VPAC(1) and VPAC(2). RO5024118 is a selective VPAC(2) receptor agonist derived via chemical modification of an earlier VPAC(2) agonist, RO0251553. In the present studies, we characterized the pharmacological activity of RO5024118.
Experimental approach: Stability of RO5024118 to human neutrophil elastase was assessed. Bronchodilatory activity of RO5024118 was investigated in guinea pig and human isolated airway smooth muscle preparations and in a guinea pig bronchoconstriction model. Pulmonary anti-inflammatory activity of RO5024118 was investigated in a lipopolysaccharide mouse model and in a porcine pancreatic elastase (PPE) rat model.
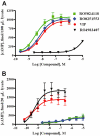
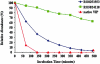
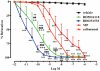
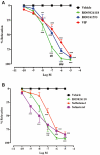
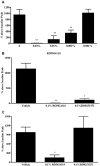
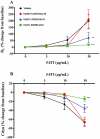
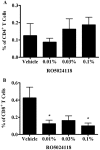
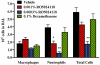
Key results: RO5024118 demonstrated increased stability to neutrophil elastase compared with RO0251553. In human and guinea pig isolated airway preparations, RO5024118 induced bronchodilatory effects comparable with RO0251553 and the long-acting β-agonist salmeterol and was significantly more potent than native vasoactive intestinal peptide and the short-acting β-agonist salbutamol. In 5-HT-induced bronchoconstriction in guinea pigs, RO5024118 exhibited inhibitory activity with similar efficacy as, and longer duration than, RO0251553. In a lipopolysaccharide-mouse model, RO5024118 inhibited neutrophil and CD8(+) cells and myeloperoxidase levels. In rats, intratracheal instillation of PPE induced airway neutrophilia that was resistant to dexamethasone. Pretreatment with RO5024118 significantly inhibited PPE-induced neutrophil accumulation.
Conclusions and implications: These results demonstrate that RO5024118 induces dual bronchodilatory and pulmonary anti-inflammatory activity and may be beneficial in treating airway obstructive and inflammatory diseases.
Linked articles: This article is part of a themed section on Analytical Receptor Pharmacology in Drug Discovery. To view the other articles in this section visit http://dx.doi.org/10.1111/bph.2010.161.issue-6.
© 2010 The Authors. British Journal of Pharmacology © 2010 The British Pharmacological Society.
Figures
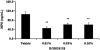

Similar articles
-
Effects of vasoactive intestinal polypeptide on antigen-induced bronchoconstriction and thromboxane release in guinea-pig lung.Br J Pharmacol. 1993 May;109(1):243-50. doi: 10.1111/j.1476-5381.1993.tb13560.x. Br J Pharmacol. 1993. PMID: 8495242 Free PMC article.
-
Ro 25-1553: a novel, long-acting vasoactive intestinal peptide agonist. Part II: Effect on in vitro and in vivo models of pulmonary anaphylaxis.J Pharmacol Exp Ther. 1994 Sep;270(3):1289-94. J Pharmacol Exp Ther. 1994. PMID: 7932181
-
Ro 25-1553: a novel, long-acting vasoactive intestinal peptide agonist. Part I: In vitro and in vivo bronchodilator studies.J Pharmacol Exp Ther. 1994 Sep;270(3):1282-8. J Pharmacol Exp Ther. 1994. PMID: 7932180
-
Novel concepts of neuropeptide-based drug therapy: vasoactive intestinal polypeptide and its receptors.Eur J Pharmacol. 2006 Mar 8;533(1-3):182-94. doi: 10.1016/j.ejphar.2005.12.055. Epub 2006 Feb 10. Eur J Pharmacol. 2006. PMID: 16473346 Review.
-
Therapeutic potential of vasoactive intestinal peptide and its receptor VPAC2 in type 2 diabetes.Front Endocrinol (Lausanne). 2022 Sep 20;13:984198. doi: 10.3389/fendo.2022.984198. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36204104 Free PMC article. Review.
Cited by
-
Beclomethasone dipropionate, formoterol fumarate and glycopyrronium bromide: Synergy of triple combination therapy on human airway smooth muscle ex vivo.Br J Pharmacol. 2020 Mar;177(5):1150-1163. doi: 10.1111/bph.14909. Epub 2020 Jan 29. Br J Pharmacol. 2020. PMID: 31660611 Free PMC article.
-
Vasodilatory effect of the stable vasoactive intestinal peptide analog RO 25-1553 in murine and rat lungs.PLoS One. 2013 Sep 19;8(9):e75861. doi: 10.1371/journal.pone.0075861. eCollection 2013. PLoS One. 2013. PMID: 24069452 Free PMC article.
References
-
- Barnes PJ. Inhaled corticosteroids are not beneficial in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2000;161:342–344. - PubMed
-
- Barnes PJ. Mediators of chronic obstructive pulmonary disease. Pharmacol Rev. 2004;56:515–548. - PubMed
-
- Battram C, Charlton SJ, Cuenoud B, Dowling MR, Fairhurst RA, Fozard JR, et al. In Vitro and in vivo pharmacological characterization of 5-[(R)-2-(5,6-diethyl-indan-2-ylamino)-1-hydroxy ethyl]-8-hydroxy-1hquinolin- 2-one (indacaterol), a novel inhaled β2 adrenoceptor agonist with a 24-h duration of action. J Pharmacol Exp Ther. 2006;317:762–770. - PubMed
-
- Belvisi MG, Hele DJ, Birrell MA. New anti-inflammatory therapies and targets for asthma and chronic obstructive pulmonary disease. Expert Opin Ther Targets. 2004;8:265–285. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

