Hemoglobin and iron handling in brain after subarachnoid hemorrhage and the effect of deferoxamine on early brain injury
- PMID: 20736956
- PMCID: PMC2970675
- DOI: 10.1038/jcbfm.2010.137
Hemoglobin and iron handling in brain after subarachnoid hemorrhage and the effect of deferoxamine on early brain injury
Abstract
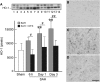
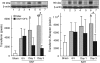
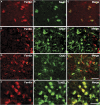
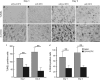
The purpose of this study was to investigate hemoglobin and iron handling after subarachnoid hemorrhage (SAH), examine the relationship between iron and neuroglial cell changes, and determine whether deferoxamine (DFX) can reduce SAH-induced injury. The SAH was induced in Sprague-Dawley rats (n=110) using an endovascular perforation technique. Animals were treated with DFX (100 mg/kg) or vehicle 2 and 6 hours after SAH induction followed by every 12 hours for 3 days. Rats were killed at 6 hours, Days 1 and 3 to determine nonheme iron and examine iron-handling proteins using Western blot and immunohistochemistry. 8-Hydroxyl-2'-deoxyguanosine and terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling (TUNEL) staining were performed to assess oxidative DNA damage and neuronal cell death. After SAH, marked heme-oxygenase-1 (HO-1) upregulation at Day 3 (P<0.01) was accompanied by elevated nonheme iron (P<0.01), transferrin (Tf) (P<0.01), Tf receptor (P<0.05), and ferritin levels (P<0.01). Deferoxamine treatment reduced SAH-induced mortality (12% versus 29%, P<0.05), brain nonheme iron concentration, iron-handling protein expression, oxidative stress, and neuronal cell death at Day 3 (P<0.01) after SAH. These results suggest that iron overload in the acute phase of SAH causes oxidative injury leading to neuronal cell death. Deferoxamine effectively reduced oxidative stress and neuronal cell death, and may be a potential therapeutic agent for SAH.
Figures



Comment in
-
Iron and early brain injury after subarachnoid hemorrhage.J Cereb Blood Flow Metab. 2010 Nov;30(11):1791-2. doi: 10.1038/jcbfm.2010.139. Epub 2010 Aug 25. J Cereb Blood Flow Metab. 2010. PMID: 20736954 Free PMC article.
References
-
- Benarroch EE. Brain iron homeostasis and neurodegenerative disease. Neurology. 2009;72:1436–1440. - PubMed
-
- Bishop GM, Robinson SR. Quantitative analysis of cell death and ferritin expression in response to cortical iron: implications for hypoxia-ischemia and stroke. Brain Res. 2001;907:175–187. - PubMed
-
- Broderick JP, Brott TG, Duldner JE, Tomsick T, Leach A. Initial and recurrent bleeding are the major causes of death following subarachnoid hemorrhage. Stroke. 1994;25:1342–1347. - PubMed
-
- Brouwers PJ, Dippel DW, Vermeulen M, Lindsay KW, Hasan D, van Gijn J. Amount of blood on computed tomography as an independent predictor after aneurysm rupture. Stroke. 1993;24:809–814. - PubMed
-
- Carbonell T, Rama R. Iron, oxidative stress and early neurological deterioration in ischemic stroke. Curr Med Chem. 2007;14:857–874. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

