Spinal injection of TNF-α-activated astrocytes produces persistent pain symptom mechanical allodynia by releasing monocyte chemoattractant protein-1
- PMID: 20737477
- PMCID: PMC2951489
- DOI: 10.1002/glia.21056
Spinal injection of TNF-α-activated astrocytes produces persistent pain symptom mechanical allodynia by releasing monocyte chemoattractant protein-1
Abstract
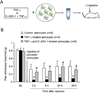
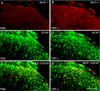
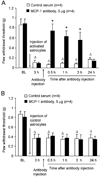
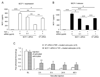
Accumulating evidence suggests that spinal astrocytes play an important role in the genesis of persistent pain, by increasing the activity of spinal cord nociceptive neurons, i.e., central sensitization. However, direct evidence of whether activation of astrocytes is sufficient to induce chronic pain symptoms is lacking. We investigated whether and how spinal injection of activated astrocytes could produce mechanical allodynia, a cardinal feature of chronic pain, in naïve mice. Spinal (intrathecal) injection of astrocytes, which were prepared from cerebral cortexes of neonatal mice and briefly stimulated by tumor necrosis factor-alpha (TNF-α), induced a substantial decrease in paw withdrawal thresholds, indicating the development of mechanical allodynia. This allodynia was prevented when the astrocyte cultures were pretreated with a peptide inhibitor of c-Jun N-terminal kinase (JNK), D-JNKI-1. Of note a short exposure of astrocytes to TNF-α for 15 min dramatically increased the expression and release of the chemokine monocyte chemoattractant protein-1 (MCP-1), even 3 h after TNF-α withdrawal, in a JNK-dependent manner. In parallel, intrathecal administration of TNF-α induced MCP-1 expression in spinal cord astrocytes. In particular, mechanical allodynia induced by TNF-α-activated astrocytes was reversed by a MCP-1 neutralizing antibody. Finally, pretreatment of astrocytes with MCP-1 siRNA attenuated astrocytes-induced mechanical allodynia. Taken together, our results suggest that activated astrocytes are sufficient to produce persistent pain symptom in naïve mice by releasing MCP-1.
© 2010 Wiley-Liss, Inc.
Figures


Similar articles
-
JNK-induced MCP-1 production in spinal cord astrocytes contributes to central sensitization and neuropathic pain.J Neurosci. 2009 Apr 1;29(13):4096-108. doi: 10.1523/JNEUROSCI.3623-08.2009. J Neurosci. 2009. PMID: 19339605 Free PMC article.
-
TRAF6 upregulation in spinal astrocytes maintains neuropathic pain by integrating TNF-α and IL-1β signaling.Pain. 2014 Dec;155(12):2618-2629. doi: 10.1016/j.pain.2014.09.027. Epub 2014 Oct 19. Pain. 2014. PMID: 25267210 Free PMC article.
-
The c-Jun N-terminal kinase 1 (JNK1) in spinal astrocytes is required for the maintenance of bilateral mechanical allodynia under a persistent inflammatory pain condition.Pain. 2010 Feb;148(2):309-319. doi: 10.1016/j.pain.2009.11.017. Pain. 2010. PMID: 20022176 Free PMC article.
-
Targeting astrocyte signaling for chronic pain.Neurotherapeutics. 2010 Oct;7(4):482-93. doi: 10.1016/j.nurt.2010.05.016. Neurotherapeutics. 2010. PMID: 20880510 Free PMC article. Review.
-
Activation of JNK pathway in persistent pain.Neurosci Lett. 2008 Jun 6;437(3):180-3. doi: 10.1016/j.neulet.2008.03.017. Epub 2008 Mar 13. Neurosci Lett. 2008. PMID: 18455869 Free PMC article. Review.
Cited by
-
P2X7 receptor induces microglia polarization to the M1 phenotype in cancer-induced bone pain rat models.Mol Pain. 2022 Jan-Dec;18:17448069211060962. doi: 10.1177/17448069211060962. Mol Pain. 2022. PMID: 35057643 Free PMC article.
-
Neuropathic pain and cytokines: current perspectives.J Pain Res. 2013 Nov 21;6:803-14. doi: 10.2147/JPR.S53660. eCollection 2013. J Pain Res. 2013. PMID: 24294006 Free PMC article. Review.
-
Levo-Tetrahydropalmatine Attenuates Bone Cancer Pain by Inhibiting Microglial Cells Activation.Mediators Inflamm. 2015;2015:752512. doi: 10.1155/2015/752512. Epub 2015 Dec 27. Mediators Inflamm. 2015. PMID: 26819501 Free PMC article.
-
Vimentin Promotes Astrocyte Activation After Chronic Constriction Injury.J Mol Neurosci. 2017 Sep;63(1):91-99. doi: 10.1007/s12031-017-0961-6. Epub 2017 Aug 8. J Mol Neurosci. 2017. PMID: 28791619
-
Chemokine signaling involving chemokine (C-C motif) ligand 2 plays a role in descending pain facilitation.Neurosci Bull. 2012 Apr;28(2):193-207. doi: 10.1007/s12264-012-1218-6. Neurosci Bull. 2012. PMID: 22466130 Free PMC article.
References
-
- Baron R. Neuropathic pain: a clinical perspective. Handb Exp Pharmacol. 2009:3–30. - PubMed
-
- Borsello T, Clarke PG, Hirt L, Vercelli A, Repici M, Schorderet DF, Bogousslavsky J, Bonny C. A peptide inhibitor of c-Jun N-terminal kinase protects against excitotoxicity and cerebral ischemia. Nat Med. 2003;9:1180–1186. - PubMed
-
- Campbell JN, Raja SN, Meyer RA, Mackinnon SE. Myelinated afferents signal the hyperalgesia associated with nerve injury. Pain. 1988;32:89–94. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

