ATP-binding cassette transporter A1 mediates the beneficial effects of the liver X receptor agonist GW3965 on object recognition memory and amyloid burden in amyloid precursor protein/presenilin 1 mice
- PMID: 20739291
- PMCID: PMC2962513
- DOI: 10.1074/jbc.M110.108100
ATP-binding cassette transporter A1 mediates the beneficial effects of the liver X receptor agonist GW3965 on object recognition memory and amyloid burden in amyloid precursor protein/presenilin 1 mice
Abstract
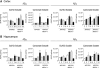
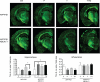
The cholesterol transporter ATP-binding cassette transporter A1 (ABCA1) moves lipids onto apolipoproteins including apolipoprotein E (apoE), which is the major cholesterol carrier in the brain and an established genetic risk factor for late-onset Alzheimer disease (AD). In amyloid mouse models of AD, ABCA1 deficiency exacerbates amyloidogenesis, whereas ABCA1 overexpression ameliorates amyloid load, suggesting a role for ABCA1 in Aβ metabolism. Agonists of liver X receptors (LXR), including GW3965, induce transcription of several genes including ABCA1 and apoE, and reduce Aβ levels and improve cognition in AD mice. However, the specific role of ABCA1 in mediating beneficial responses to LXR agonists in AD mice is unknown. We evaluated behavioral and neuropathogical outcomes in GW3965-treated female APP/PS1 mice with and without ABCA1. Treatment of APP/PS1 mice with GW3965 increased ABCA1 and apoE protein levels. ABCA1 was required to observe significantly elevated apoE levels in brain tissue and cerebrospinal fluid upon therapeutic (33 mg/kg/day) GW3965 treatment. At 33 mg/kg/day, GW3965 was also associated with a trend toward redistribution of Aβ to the carbonate-soluble pool independent of ABCA1. APP/PS1 mice treated with either 2.5 or 33 mg/kg/day of GW3965 showed a clear trend toward reduced amyloid burden in hippocampus and whole brain, whereas APP/PS1-treated mice lacking ABCA1 failed to display reduced amyloid load in the whole brain and showed trends toward increased hippocampal amyloid. Treatment of APP/PS1 mice with either dose of GW3965 completely restored novel object recognition memory to wild-type levels, which required ABCA1. These results suggest that ABCA1 contributes to several beneficial effects of the LXR agonist GW3965 in APP/PS1 mice.
Figures




References
-
- Fan J., Donkin J., Wellington C. L. (2009) Biofactors 35, 239–248 - PubMed
-
- Morris J. C. (1997) Neurology 49, (suppl.) S7–S10 - PubMed
-
- Hardy J. (1997) Trends Neurosci. 20, 154–159 - PubMed
-
- Bates K. A., Verdile G., Li Q. X., Ames D., Hudson P., Masters C. L., Martins R. N. (2009) Mol. Psychiatry 14, 469–486 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

