Hippocampal ripple-contingent training accelerates trace eyeblink conditioning and retards extinction in rabbits
- PMID: 20739570
- PMCID: PMC6633352
- DOI: 10.1523/JNEUROSCI.2165-10.2010
Hippocampal ripple-contingent training accelerates trace eyeblink conditioning and retards extinction in rabbits
Abstract
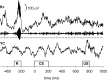
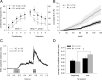
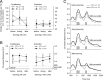
There are at least two distinct oscillatory states of the hippocampus that are related to distinct behavioral patterns. Theta (4-12 Hz) oscillation has been suggested to indicate selective attention during which the animal concentrates on some features of the environment while suppressing reactivity to others. In contrast, sharp-wave ripples ( approximately 200 Hz) can be seen in a state in which the hippocampus is at its most responsive to any kind of afferent stimulation. In addition, external stimulation tends to evoke and reset theta oscillation, the phase of which has been shown to modulate synaptic plasticity in the hippocampus. Theoretically, training on a hippocampus-dependent learning task contingent upon ripples could enhance learning rate due to elevated responsiveness and enhanced phase locking of the theta oscillation. We used a brain-computer interface to detect hippocampal ripples in rabbits to deliver trace eyeblink conditioning and extinction trials selectively contingent upon them. A yoked control group was trained regardless of their ongoing neural state. Ripple-contingent training expedited acquisition of the conditioned response early in training and evoked stronger theta-band phase locking to the conditioned stimulus. Surprisingly, ripple-contingent training also resulted in slower extinction in well trained animals. We suggest that the ongoing oscillatory activity in the hippocampus determines the extent to which a stimulus can induce a phase reset of the theta oscillation, which in turn is the determining factor of learning rate in trace eyeblink conditioning.
Figures

Similar articles
-
Effects of hippocampal state-contingent trial presentation on hippocampus-dependent nonspatial classical conditioning and extinction.J Neurosci. 2014 Apr 23;34(17):6003-10. doi: 10.1523/JNEUROSCI.4859-13.2014. J Neurosci. 2014. PMID: 24760859 Free PMC article.
-
Hippocampal theta phase-contingent memory retrieval in delay and trace eyeblink conditioning.Behav Brain Res. 2018 Jan 30;337:264-270. doi: 10.1016/j.bbr.2017.09.001. Epub 2017 Sep 4. Behav Brain Res. 2018. PMID: 28882692
-
Hippocampal theta-band activity and trace eyeblink conditioning in rabbits.Behav Neurosci. 2009 Jun;123(3):631-40. doi: 10.1037/a0015334. Behav Neurosci. 2009. PMID: 19485570
-
Brain mechanisms of extinction of the classically conditioned eyeblink response.Learn Mem. 2004 Sep-Oct;11(5):517-24. doi: 10.1101/lm.80004. Learn Mem. 2004. PMID: 15466302 Review.
-
Harnessing the power of theta: natural manipulations of cognitive performance during hippocampal theta-contingent eyeblink conditioning.Front Syst Neurosci. 2015 Apr 13;9:50. doi: 10.3389/fnsys.2015.00050. eCollection 2015. Front Syst Neurosci. 2015. PMID: 25918501 Free PMC article. Review.
Cited by
-
Real-time classification of experience-related ensemble spiking patterns for closed-loop applications.Elife. 2018 Oct 30;7:e36275. doi: 10.7554/eLife.36275. Elife. 2018. PMID: 30373716 Free PMC article.
-
The hippocampal sharp wave-ripple in memory retrieval for immediate use and consolidation.Nat Rev Neurosci. 2018 Dec;19(12):744-757. doi: 10.1038/s41583-018-0077-1. Nat Rev Neurosci. 2018. PMID: 30356103 Free PMC article. Review.
-
Spatial tuning and brain state account for dorsal hippocampal CA1 activity in a non-spatial learning task.Elife. 2016 Aug 3;5:e14321. doi: 10.7554/eLife.14321. Elife. 2016. PMID: 27487561 Free PMC article.
-
Project DyAdd: classical eyeblink conditioning in adults with dyslexia and ADHD.Exp Brain Res. 2012 Nov;223(1):19-32. doi: 10.1007/s00221-012-3237-y. Epub 2012 Sep 5. Exp Brain Res. 2012. PMID: 22948736
-
Scaling brain size, keeping timing: evolutionary preservation of brain rhythms.Neuron. 2013 Oct 30;80(3):751-64. doi: 10.1016/j.neuron.2013.10.002. Neuron. 2013. PMID: 24183025 Free PMC article. Review.
References
-
- Bures J, Petran M, Zachar J. Electrophysiological methods in biological research. New York: Academic; 1967.
-
- Buzsáki G. Hippocampal sharp waves: their origin and significance. Brain Res. 1986;398:242–252. - PubMed
-
- Buzsáki G. Two-stage model of memory trace formation: a role for “noisy” brain states. Neuroscience. 1989;31:551–570. - PubMed
-
- Buzsáki G. Theta oscillations in the hippocampus. Neuron. 2002;33:325–340. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
