Should animals navigating over short distances switch to a magnetic compass sense?
- PMID: 20740070
- PMCID: PMC2927193
- DOI: 10.3389/fnbeh.2010.00042
Should animals navigating over short distances switch to a magnetic compass sense?
Abstract
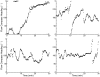
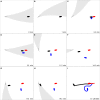
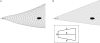
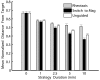
Magnetoreception can play a substantial role in long distance navigation by animals. I hypothesize that locomotion guided by a magnetic compass sense could also play a role in short distance navigation. Animals identify mates, prey, or other short distance navigational goals using different sensory modalities (olfaction, vision, audition, etc.) to detect sensory cues associated with those goals. In conditions where these cues become unreliable for navigation (due to flow changes, obstructions, noise interference, etc.), switching to a magnetic compass sense to guide locomotion toward the navigational goals could be beneficial. Using simulations based on known locomotory and flow parameters, I show this strategy has strong theoretical benefits for the nudibranch mollusk Tritonia diomedea navigating toward odor sources in variable flow. A number of other animals may garner similar benefits, particularly slow-moving species in environments with rapidly changing cues relevant for navigation. Faster animals might also benefit from switching to a magnetic compass sense, provided the initial cues used for navigation (acoustic signals, odors, etc.) are intermittent or change rapidly enough that the entire navigation behavior cannot be guided by a continuously detectable cue. Examination of the relative durations of navigational tasks, the persistence of navigational cues, and the stability of both navigators and navigational targets will identify candidates with the appropriate combination of unreliable initial cues and relatively immobile navigational goals for which this hypothetical behavior could be beneficial. Magnetic manipulations can then test whether a switch to a magnetic compass sense occurs. This hypothesis thus provides an alternative when considering the behavioral significance of a magnetic compass sense in animals.
Keywords: Tritonia; acoustic signaling; magnetic compass sense; magnetoreception; navigation; odor-gated rheotaxis; short distance; simulation.
Figures
Similar articles
-
Magnetoreception in Hymenoptera: importance for navigation.Anim Cogn. 2020 Nov;23(6):1051-1061. doi: 10.1007/s10071-020-01431-x. Epub 2020 Sep 25. Anim Cogn. 2020. PMID: 32975654 Free PMC article. Review.
-
Additional Navigational Strategies Can Augment Odor-Gated Rheotaxis for Navigation under Conditions of Variable Flow.Integr Comp Biol. 2015 Sep;55(3):447-60. doi: 10.1093/icb/icv073. Epub 2015 Jun 26. Integr Comp Biol. 2015. PMID: 26116202
-
One rhinophore probably provides sufficient sensory input for odour-based navigation by the nudibranch mollusc Tritonia diomedea.J Exp Biol. 2014 Dec 1;217(Pt 23):4149-58. doi: 10.1242/jeb.111153. Epub 2014 Oct 16. J Exp Biol. 2014. PMID: 25324338
-
The Neural Basis of Long-Distance Navigation in Birds.Annu Rev Physiol. 2016;78:133-54. doi: 10.1146/annurev-physiol-021115-105054. Epub 2015 Nov 2. Annu Rev Physiol. 2016. PMID: 26527184 Review.
-
Orientation and navigation relative to water flow, prey, conspecifics, and predators by the nudibranch mollusc Tritonia diomedea.Biol Bull. 2006 Apr;210(2):97-108. doi: 10.2307/4134599. Biol Bull. 2006. PMID: 16641515
Cited by
-
Magnetosensation during re-learning walks in desert ants (Cataglyphis nodus).J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2022 Jan;208(1):125-133. doi: 10.1007/s00359-021-01511-4. Epub 2021 Oct 22. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2022. PMID: 34677637 Free PMC article.
-
How Swift Is Cry-Mediated Magnetoreception? Conditioning in an American Cockroach Shows Sub-second Response.Front Behav Neurosci. 2018 May 28;12:107. doi: 10.3389/fnbeh.2018.00107. eCollection 2018. Front Behav Neurosci. 2018. PMID: 29892217 Free PMC article.
-
Magnetoreception in Hymenoptera: importance for navigation.Anim Cogn. 2020 Nov;23(6):1051-1061. doi: 10.1007/s10071-020-01431-x. Epub 2020 Sep 25. Anim Cogn. 2020. PMID: 32975654 Free PMC article. Review.
References
-
- Arendse M. C. (1978). Magnetic field detection is distinct from light detection in the invertebrates Tenebrio and Talitrus. Nature 274, 358–36210.1038/274358a0 - DOI
-
- Birkeland C. (1974). Interactions between a sea pen and seven of its predators. Ecol. Monogr. 44, 211–23210.2307/1942312 - DOI
-
- Bonadonna F., Bajzak C., Benhamou S., Igloi K., Jouventin P., Lipp H. P., Dell'Omo G. (2005). Orientation in the wandering albatross: interfering with magnetic perception does not affect orientation performance. Proc. R. Soc. Lond., B, Biol. Sci. 272, 489–49510.1098/rspb.2004.2984 - DOI - PMC - PubMed
-
- Bonadonna F., Spaggiari J., Weimerskirch H. (2001). Could osmotaxis explain the ability of blue petrels to return to their burrows at night? J. Exp. Biol. 204, 1485–1489 - PubMed
LinkOut - more resources
Full Text Sources

