Characterization of the interaction of β-amyloid with transthyretin monomers and tetramers
- PMID: 20795734
- PMCID: PMC2943652
- DOI: 10.1021/bi101280t
Characterization of the interaction of β-amyloid with transthyretin monomers and tetramers
Abstract
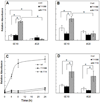
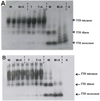
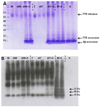
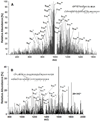
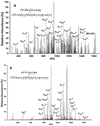
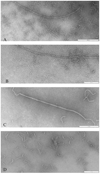
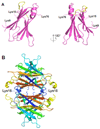
β-Amyloid (Aβ) is the main protein component of the amyloid plaques associated with Alzheimer's disease. Transthyretin (TTR) is a homotetramer that circulates in both blood and cerebrospinal fluid. Wild-type (wt) TTR amyloid deposits are linked to senile systemic amyloidosis, a common disease of aging, while several TTR mutants are linked to familial amyloid polyneuropathy. Several recent studies provide support for the hypothesis that these two amyloidogenic proteins interact, and that this interaction is biologically relevant. For example, upregulation of TTR expression in Tg2576 mice was linked to protection from the toxic effects of Aβ deposition [Stein, T. D., and Johnson, J. A. (2002) J. Neurosci. 22, 7380-7388]. We examined the interaction of Aβ with wt TTR as well as two mutants: F87M/L110M, engineered to be a stable monomer, and T119M, a naturally occurring mutant with a tetrameric stability higher than that of the wild type. On the basis of enzyme-linked immunoassays as well as cross-linking experiments, we conclude that Aβ monomers bind more to TTR monomers than to TTR tetramers. The data further suggest that TTR tetramers interact preferably with Aβ aggregates rather than Aβ monomers. Through tandem mass spectrometry analysis of cross-linked TTR-Aβ fragments, we identified the A strand, in the inner β-sheet of TTR, as well as the EF helix, as regions of TTR that are involved with Aβ association. Light scattering and electron microscopy studies demonstrate that the outcome of the TTR-Aβ interaction strongly depends on TTR quaternary structure. While TTR tetramers may modestly enhance aggregation, TTR monomers decidedly arrest Aβ aggregate growth. These data provide important new insights into the nature of TTR-Aβ interactions. Such interactions may regulate TTR-mediated protection against Aβ toxicity.
Figures






Similar articles
-
Transthyretin and the brain re-visited: is neuronal synthesis of transthyretin protective in Alzheimer's disease?Mol Neurodegener. 2011 Nov 23;6:79. doi: 10.1186/1750-1326-6-79. Mol Neurodegener. 2011. PMID: 22112803 Free PMC article. Review.
-
Transthyretin as both a sensor and a scavenger of β-amyloid oligomers.Biochemistry. 2013 Apr 30;52(17):2849-61. doi: 10.1021/bi4001613. Epub 2013 Apr 19. Biochemistry. 2013. PMID: 23570378 Free PMC article.
-
Trapping the monomer of a non-amyloidogenic variant of transthyretin: exploring its possible use as a therapeutic strategy against transthyretin amyloidogenic diseases.J Biol Chem. 2009 Jan 16;284(3):1443-53. doi: 10.1074/jbc.M807100200. Epub 2008 Nov 4. J Biol Chem. 2009. PMID: 18984591
-
Inhibition of amyloid beta fibril formation by monomeric human transthyretin.Protein Sci. 2018 Jul;27(7):1252-1261. doi: 10.1002/pro.3396. Epub 2018 Mar 14. Protein Sci. 2018. PMID: 29498118 Free PMC article.
-
Nearly 200 X-ray crystal structures of transthyretin: what do they tell us about this protein and the design of drugs for TTR amyloidoses?Curr Med Chem. 2012;19(15):2324-42. doi: 10.2174/092986712800269335. Curr Med Chem. 2012. PMID: 22471981 Review.
Cited by
-
Pathology of Amyloid-β (Aβ) Peptide Peripheral Clearance in Alzheimer's Disease.Int J Mol Sci. 2024 Oct 11;25(20):10964. doi: 10.3390/ijms252010964. Int J Mol Sci. 2024. PMID: 39456746 Free PMC article. Review.
-
Identification of subunit-subunit interaction sites in αA-WT crystallin and mutant αA-G98R crystallin using isotope-labeled cross-linker and mass spectrometry.PLoS One. 2013 Jun 5;8(6):e65610. doi: 10.1371/journal.pone.0065610. Print 2013. PLoS One. 2013. PMID: 23755258 Free PMC article.
-
Approaches for Increasing Cerebral Efflux of Amyloid-β in Experimental Systems.J Alzheimers Dis. 2024;100(2):379-411. doi: 10.3233/JAD-240212. J Alzheimers Dis. 2024. PMID: 38875041 Free PMC article. Review.
-
Transthyretin and the brain re-visited: is neuronal synthesis of transthyretin protective in Alzheimer's disease?Mol Neurodegener. 2011 Nov 23;6:79. doi: 10.1186/1750-1326-6-79. Mol Neurodegener. 2011. PMID: 22112803 Free PMC article. Review.
-
Identification of beta-amyloid-binding sites on transthyretin.Protein Eng Des Sel. 2012 Jul;25(7):337-45. doi: 10.1093/protein/gzs026. Epub 2012 Jun 4. Protein Eng Des Sel. 2012. PMID: 22670059 Free PMC article.
References
-
- Gralle M, Ferreira ST. Structure and functions of the human amyloid precursor protein: The whole is more than the sum of its parts. Prog Neurobiol. 2007;82:11–32. - PubMed
-
- Hardy J. Testing times for the "amyloid cascade hypothesis". Neurobiol Aging. 2002;23:1073–1074. - PubMed
-
- Wirths O, Multhaup G, Bayer TA. A modified beta-amyloid hypothesis: intraneuronal accumulation of the beta-amyloid peptide - the first step of a fatal cascade. J Neurochem. 2004;91:513–520. - PubMed
-
- Lee HG, Casadesus G, Zhu XW, Takeda A, Perry G, Smith MA. Challenging the amyloid cascade hypothesis - Senile plaques and amyloid-beta as protective adaptations to Alzheimer disease. Strategies for Engineered Negligible Senescence: Why Genuine Control of Aging May Be Foreseeable. 2004;1019:1–4. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

