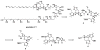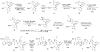Synthesis of the bis-tetrahydropyran core of amphidinol 3
- PMID: 20795745
- PMCID: PMC2929927
- DOI: 10.1021/ol1015898
Synthesis of the bis-tetrahydropyran core of amphidinol 3
Abstract
A convergent synthesis of the C31-C52 bis-tetrahydropyran core of the natural product amphidinol 3 is reported. A common intermediate was synthesized from d-tartaric acid utilizing an asymmetric glycolate alkylation/ring-closing metathesis sequence to construct the THP rings. Differential elaboration of the common intermediate allowed the synthesis of two distinct coupling partners which were joined through a modified Horner-Wadsworth-Emmons olefination to provide the bis-tetrahydropyran core.
Figures
Similar articles
-
Development of an access route to the c31-c52 central core of amphidinol 3.Org Lett. 2007 Feb 1;9(3):513-6. doi: 10.1021/ol062875+. Org Lett. 2007. PMID: 17249800
-
Stereoselective synthesis of the C31-C40/C43-C52 unit of amphidinol 3.J Org Chem. 2009 Nov 20;74(22):8810-3. doi: 10.1021/jo901793f. J Org Chem. 2009. PMID: 19852459
-
Synthesis and Stereochemical Revision of the C31-C67 Fragment of Amphidinol 3.Angew Chem Int Ed Engl. 2018 May 22;57(21):6060-6064. doi: 10.1002/anie.201712167. Epub 2018 Apr 27. Angew Chem Int Ed Engl. 2018. PMID: 29635773
-
Strategies for the construction of tetrahydropyran rings in the synthesis of natural products.Org Biomol Chem. 2014 Jun 7;12(21):3323-35. doi: 10.1039/c4ob00423j. Epub 2014 Apr 17. Org Biomol Chem. 2014. PMID: 24744139 Review.
-
SmI2-induced cyclizations and their applications in natural product synthesis.Chem Rec. 2010 Jun;10(3):159-72. doi: 10.1002/tcr.200900027. Chem Rec. 2010. PMID: 20503205 Review.
Cited by
-
Karlotoxin synthetic studies: concise synthesis of a C(42-63) B-ring tetrahydropyran fragment.Tetrahedron Lett. 2013 Nov 1;54(48):10.1016/j.tetlet.2013.09.104. doi: 10.1016/j.tetlet.2013.09.104. Tetrahedron Lett. 2013. PMID: 24376284 Free PMC article.
-
A highly convergent synthesis of the C1-C31 polyol domain of amphidinol 3 featuring a TST-RCM reaction: confirmation of the revised relative stereochemistry.Chem Sci. 2015 Nov 1;6(11):6407-6412. doi: 10.1039/c5sc00814j. Epub 2015 Aug 6. Chem Sci. 2015. PMID: 28757956 Free PMC article.
-
INTEGRATED APPROACHES TO THE CONFIGURATIONAL ASSIGNMENT OF MARINE NATURAL PRODUCTS.Tetrahedron. 2012 Nov 18;68(46):9307-9343. doi: 10.1016/j.tet.2011.12.070. Tetrahedron. 2012. PMID: 23814320 Free PMC article. No abstract available.
-
All-Heteroatom-Substituted Carbon Spiro Stereocenters: Synthesis, Resolution, Enantiomeric Stability, and Absolute Configuration.J Am Chem Soc. 2025 Jun 18;147(24):21121-21130. doi: 10.1021/jacs.5c06394. Epub 2025 Jun 4. J Am Chem Soc. 2025. PMID: 40464057 Free PMC article.
References
-
- Satake M, Murata M, Yasumoto T, Fujita T, Naoki H. J Am Chem Soc. 1991;113:9859–9861.
-
- Paul GK, Matsumori N, Konoki K, Sasaki M, Murata M, Tachibana K. Harmful and Toxic Algal Blooms
-
- Murata M, Matsuoka S, Matsumori N, Paul GK, Tachibana K. J Am Chem Soc. 1999;121:870.
-
- Oishi T, Kanemoto M, Swasono R, Matsumori N, Murata M. Org Lett. 2008;10:5203–5206. - PubMed
-
- BouzBouz S, Cossy J. Org Lett. 2001;3:1451–1454. - PubMed
- Cossy J, Tsuchiya T, Ferrie L, Reymond S, Kreuzer T, Colobert F, Jourdain P, Marko IE. Synlett. 2007:2286–2288.
- Colobert F, Kreuzer T, Cossy J, Reymond S, Tsuchiya T, Ferrie L, Marko IE, Jourdain P. Synlett. 2007:2351–2354.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous