The PI3K/Akt pathway upregulates Id1 and integrin α4 to enhance recruitment of human ovarian cancer endothelial progenitor cells
- PMID: 20796276
- PMCID: PMC2940800
- DOI: 10.1186/1471-2407-10-459
The PI3K/Akt pathway upregulates Id1 and integrin α4 to enhance recruitment of human ovarian cancer endothelial progenitor cells
Abstract
Background: Endothelial progenitor cells (EPCs) contribute to tumor angiogenesis and growth. We aimed to determine whether inhibitors of differentiation 1 (Id1) were expressed in circulating EPCs of patients with ovarian cancer, whether Id1 could mediate EPCs mobilization and recruitment, and, if so, what underlying signaling pathway it used.
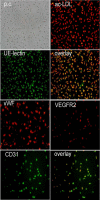
Methods: Circulating EPCs cultures were from 25 patients with ovarian cancer and 20 healthy control subjects. Id1 and integrin α4 expression were analyzed by real-time reverse transcription-polymerase chain reaction and western blot. EPCs proliferation, migration, and adhesion were detected by MTT, transwell chamber, and EPCs-matrigel adhesion assays. Double-stranded DNA containing the interference sequences were synthesized according to the structure of a pGCSIL-GFP viral vector and then inserted into a linearized vector. Positive clones were identified as lentiviral vectors that expressed human Id1 short hairpin RNA (shRNA).
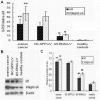
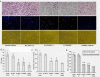
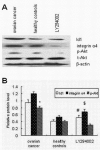
Results: Id1 and integrin α4 expression were increased in EPCs freshly isolated from ovarian cancer patients compared to those obtained from healthy subjects. siRNA-mediated Id1 downregulation substantially reduced EPCs function and integrin α4 expression. Importantly, Inhibition of PI3K/Akt inhibited Id1 and integrin α4 expression, resulting in the decreasing biological function of EPCs.
Conclusions: Id1 induced EPCs mobilization and recruitment is mediated chiefly by the PI3K/Akt signaling pathway and is associated with activation of integrin α4.
Figures
Similar articles
-
Id1 enhances human ovarian cancer endothelial progenitor cell angiogenesis via PI3K/Akt and NF-κB/MMP-2 signaling pathways.J Transl Med. 2013 May 29;11:132. doi: 10.1186/1479-5876-11-132. J Transl Med. 2013. PMID: 23714001 Free PMC article.
-
An essential role for the Id1/PI3K/Akt/NFkB/survivin signalling pathway in promoting the proliferation of endothelial progenitor cells in vitro.Mol Cell Biochem. 2012 Apr;363(1-2):135-45. doi: 10.1007/s11010-011-1166-x. Epub 2011 Dec 3. Mol Cell Biochem. 2012. PMID: 22139302 Free PMC article.
-
Follicle-stimulating hormone induced epithelial-mesenchymal transition of epithelial ovarian cancer cells through follicle-stimulating hormone receptor PI3K/Akt-Snail signaling pathway.Int J Gynecol Cancer. 2014 Nov;24(9):1564-74. doi: 10.1097/IGC.0000000000000279. Int J Gynecol Cancer. 2014. PMID: 25340291
-
Effects of metastasis-associated in colon cancer 1 inhibition by small hairpin RNA on ovarian carcinoma OVCAR-3 cells.J Exp Clin Cancer Res. 2011 Sep 16;30(1):83. doi: 10.1186/1756-9966-30-83. J Exp Clin Cancer Res. 2011. PMID: 21923915 Free PMC article.
-
Hypoxia upregulates ovarian cancer invasiveness via the binding of HIF-1α to a hypoxia-induced, methylation-free hypoxia response element of S100A4 gene.Int J Cancer. 2012 Oct 15;131(8):1755-67. doi: 10.1002/ijc.27448. Epub 2012 Mar 14. Int J Cancer. 2012. PMID: 22287060
Cited by
-
Id1 enhances human ovarian cancer endothelial progenitor cell angiogenesis via PI3K/Akt and NF-κB/MMP-2 signaling pathways.J Transl Med. 2013 May 29;11:132. doi: 10.1186/1479-5876-11-132. J Transl Med. 2013. PMID: 23714001 Free PMC article.
-
Effects of suberoylanilide hydroxamic acid (SAHA) combined with paclitaxel (PTX) on paclitaxel-resistant ovarian cancer cells and insights into the underlying mechanisms.Cancer Cell Int. 2014 Nov 26;14(1):112. doi: 10.1186/s12935-014-0112-x. eCollection 2014. Cancer Cell Int. 2014. PMID: 25546354 Free PMC article.
-
The integrin αv-TGFβ signaling axis is necessary for epidermal proliferation during cutaneous wound healing.Cell Cycle. 2016 Aug 2;15(15):2077-86. doi: 10.1080/15384101.2016.1199306. Epub 2016 Jun 13. Cell Cycle. 2016. PMID: 27295308 Free PMC article.
-
ID1 confers cancer cell chemoresistance through STAT3/ATF6-mediated induction of autophagy.Cell Death Dis. 2020 Feb 20;11(2):137. doi: 10.1038/s41419-020-2327-1. Cell Death Dis. 2020. PMID: 32080166 Free PMC article.
-
Identification of Id1 as a downstream effector for arsenic-promoted angiogenesis via PI3K/Akt, NF-κB and NOS signaling.Toxicol Res (Camb). 2015 Oct 5;5(1):151-159. doi: 10.1039/c5tx00280j. eCollection 2016 Jan 1. Toxicol Res (Camb). 2015. PMID: 30090333 Free PMC article.
References
-
- Stoelting S, Trefzer T, Kisro J, Steinke A, Wagner T, Peters SO. Low-dose oral metronomic chemotherapy prevents mobilization of endothelial progenitor cells into the blood of cancer patients. In Vivo. 2000;822(6):831–836. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

