Autocrine regulation of human sperm motility by tachykinins
- PMID: 20796280
- PMCID: PMC2936315
- DOI: 10.1186/1477-7827-8-104
Autocrine regulation of human sperm motility by tachykinins
Abstract
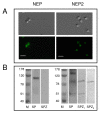
Background: We examined the presence and function of tachykinins and the tachykinin-degrading enzymes neprilysin (NEP) and neprilysin-2 (NEP2) in human spermatozoa.
Methods: Freshly ejaculated semen was collected from forty-eight normozoospermic human donors. We analyzed the expression of substance P, neurokinin A, neurokinin B, hemokinin-1, NEP and NEP2 in sperm cells by reverse-transcriptase polymerase chain reaction (RT-PCR), western blot and immunocytochemistry assays and evaluated the effects of the neprilysin and neprilysin-2 inhibitor phosphoramidon on sperm motility in the absence and presence of tachykinin receptor-selective antagonists. Sperm motility was measured using WHO procedures or computer-assisted sperm analysis (CASA).
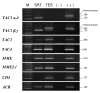
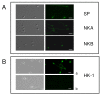
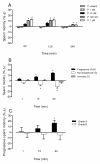
Results: The mRNAs of the genes that encode substance P/neurokinin A (TAC1), neurokinin B (TAC3), hemokinin-1 (TAC4), neprilysin (MME) and neprilysin-2 (MMEL1) were expressed in human sperm. Immunocytochemistry studies revealed that tachykinin and neprilysin proteins were present in spermatozoa and show specific and differential distributions. Phosphoramidon increased sperm progressive motility and its effects were reduced in the presence of the tachykinin receptor antagonists SR140333 (NK1 receptor-selective) and SR48968 (NK2 receptor-selective) but unmodified in the presence of SR142801 (NK3 receptor-selective).
Conclusion: These data show that tachykinins are present in human spermatozoa and participate in the regulation of sperm motility. Tachykinin activity is regulated, at least in part, by neprilysins.
Figures
Similar articles
-
A role for tachykinins in the regulation of human sperm motility.Hum Reprod. 2007 Jun;22(6):1617-25. doi: 10.1093/humrep/dem069. Epub 2007 Apr 16. Hum Reprod. 2007. PMID: 17437961
-
A role for tachykinins in female mouse and rat reproductive function.Biol Reprod. 2003 Sep;69(3):940-6. doi: 10.1095/biolreprod.103.017111. Epub 2003 May 28. Biol Reprod. 2003. PMID: 12773411
-
Functional and molecular characterization of tachykinins and tachykinin receptors in the mouse uterus.Biol Reprod. 2005 May;72(5):1125-33. doi: 10.1095/biolreprod.104.036814. Epub 2005 Jan 12. Biol Reprod. 2005. PMID: 15647454
-
Non-neuronal mammalian tachykinin expression.Front Biosci. 2004 Sep 1;9:2166-76. doi: 10.2741/1372. Front Biosci. 2004. PMID: 15353278 Review.
-
Mammalian tachykinins and uterine smooth muscle: the challenge escalates.Eur J Pharmacol. 2004 Oct 1;500(1-3):15-26. doi: 10.1016/j.ejphar.2004.07.007. Eur J Pharmacol. 2004. PMID: 15464017 Review.
Cited by
-
(Pro)renin Receptor Is Present in Human Sperm and It Adversely Affects Sperm Fertility Ability.Int J Mol Sci. 2021 Mar 22;22(6):3215. doi: 10.3390/ijms22063215. Int J Mol Sci. 2021. PMID: 33809946 Free PMC article.
-
Proteomic analysis reveals dysregulated cell signaling in ejaculated spermatozoa from infertile men.Asian J Androl. 2019 Mar-Apr;21(2):121-130. doi: 10.4103/aja.aja_56_18. Asian J Androl. 2019. PMID: 30381577 Free PMC article.
-
A comprehensive review of the literature on CD10: its function, clinical application, and prospects.Front Pharmacol. 2024 Feb 8;15:1336310. doi: 10.3389/fphar.2024.1336310. eCollection 2024. Front Pharmacol. 2024. PMID: 38389922 Free PMC article. Review.
-
IGFs Potentiate TAC3-induced SLα Expression via Upregulation of TACR3 Expression in Grass Carp Pituitary Cells.Cells. 2019 Aug 13;8(8):887. doi: 10.3390/cells8080887. Cells. 2019. PMID: 31412674 Free PMC article.
-
Regulation of Male Fertility by the Renin-Angiotensin System.Int J Mol Sci. 2020 Oct 26;21(21):7943. doi: 10.3390/ijms21217943. Int J Mol Sci. 2020. PMID: 33114706 Free PMC article. Review.
References
-
- Pinto FM, Armesto CP, Magraner J, Trujillo M, Martín JD, Candenas ML. Tachykinin receptor and neutral endopeptidase gene expression in the rat uterus: characterization and regulation in response to ovarian steroid treatment. Endocrinology. 1999;140:2526–2532. doi: 10.1210/en.140.6.2526. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

