The chemokine receptor CXCR5 is pivotal for ectopic mucosa-associated lymphoid tissue neogenesis in chronic Helicobacter pylori-induced inflammation
- PMID: 20798913
- PMCID: PMC2956061
- DOI: 10.1007/s00109-010-0658-6
The chemokine receptor CXCR5 is pivotal for ectopic mucosa-associated lymphoid tissue neogenesis in chronic Helicobacter pylori-induced inflammation
Abstract
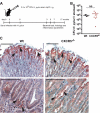
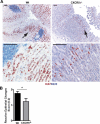
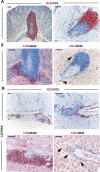
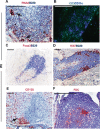
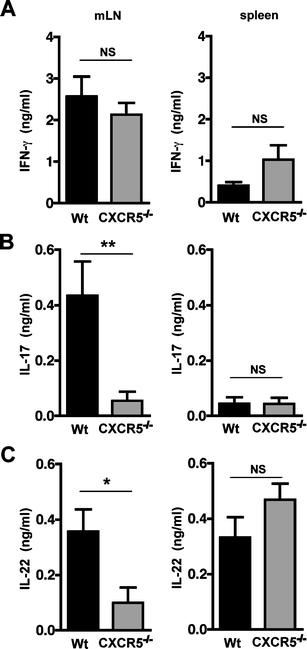
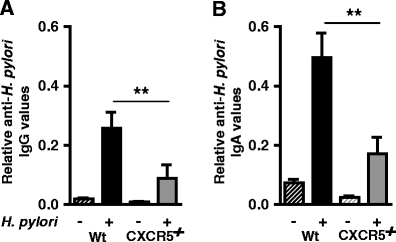
Ectopic lymphoid follicles are a key feature of chronic inflammatory autoimmune and infectious diseases, such as rheumatoid arthritis, Sjögren's syndrome, and Helicobacter pylori-induced gastritis. Homeostatic chemokines are considered to be involved in the formation of such tertiary lymphoid tissue. High expression of CXCL13 and its receptor, CXCR5, has been associated with the formation of ectopic lymphoid follicles in chronic infectious diseases. Here, we defined the role of CXCR5 in the development of mucosal tertiary lymphoid tissue and gastric inflammation in a mouse model of chronic H. pylori infection. CXCR5-deficient mice failed to develop organized gastric lymphoid follicles despite similar bacterial colonization density as infected wild-type mice. CXCR5 deficiency altered Th17 responses but not Th1-type cellular immune responses to H. pylori infection. Furthermore, CXCR5-deficient mice exhibited lower H. pylori-specific serum IgG and IgA levels and an overall decrease in chronic gastric immune responses. In conclusion, the development of mucosal tertiary ectopic follicles during chronic H. pylori infection is strongly dependent on the CXCL13/CXCR5 signaling axis, and lack of de novo lymphoid tissue formation attenuates chronic immune responses.
Figures

Similar articles
-
CXCR5- and CCR7-dependent lymphoid neogenesis in a murine model of chronic antigen-induced arthritis.Arthritis Rheum. 2007 Oct;56(10):3271-83. doi: 10.1002/art.22939. Arthritis Rheum. 2007. PMID: 17907173
-
Enhanced expression of CXCL13 in human Helicobacter pylori-associated gastritis.Dig Dis Sci. 2011 Oct;56(10):2887-94. doi: 10.1007/s10620-011-1717-8. Epub 2011 Jun 7. Dig Dis Sci. 2011. PMID: 21647655
-
BCA-1 is highly expressed in Helicobacter pylori-induced mucosa-associated lymphoid tissue and gastric lymphoma.J Clin Invest. 1999 Nov;104(10):R49-54. doi: 10.1172/JCI7830. J Clin Invest. 1999. PMID: 10562310 Free PMC article.
-
Effects of CXCL13 inhibition on lymphoid follicles in models of autoimmune disease.Eur J Clin Invest. 2013 May;43(5):501-9. doi: 10.1111/eci.12063. Epub 2013 Mar 20. Eur J Clin Invest. 2013. PMID: 23517338 Review.
-
Role of the CXCL13/CXCR5 Axis in Autoimmune Diseases.Front Immunol. 2022 Mar 4;13:850998. doi: 10.3389/fimmu.2022.850998. eCollection 2022. Front Immunol. 2022. PMID: 35309354 Free PMC article. Review.
Cited by
-
Tertiary lymphoid structures are associated with higher tumor grade in primary operable breast cancer patients.BMC Cancer. 2015 Mar 6;15:101. doi: 10.1186/s12885-015-1116-1. BMC Cancer. 2015. PMID: 25884667 Free PMC article.
-
Helicobacter pylori and its association with autoimmune diseases: systemic lupus erythematosus, rheumatoid arthritis and Sjögren syndrome.J Transl Autoimmun. 2021 Nov 14;4:100135. doi: 10.1016/j.jtauto.2021.100135. eCollection 2021. J Transl Autoimmun. 2021. PMID: 34825158 Free PMC article.
-
High endothelial venules (HEVs) in immunity, inflammation and cancer.Angiogenesis. 2021 Nov;24(4):719-753. doi: 10.1007/s10456-021-09792-8. Epub 2021 May 6. Angiogenesis. 2021. PMID: 33956259 Free PMC article. Review.
-
Anti-CXCL13 antibody can inhibit the formation of gastric lymphoid follicles induced by Helicobacter infection.Mucosal Immunol. 2014 Sep;7(5):1244-54. doi: 10.1038/mi.2014.14. Epub 2014 Mar 19. Mucosal Immunol. 2014. PMID: 24646940
-
Adventitial inflammation and its interaction with intimal atherosclerotic lesions.Front Physiol. 2014 Aug 8;5:296. doi: 10.3389/fphys.2014.00296. eCollection 2014. Front Physiol. 2014. PMID: 25152736 Free PMC article. Review.
References
-
- Manzo A, Paoletti S, Carulli M, Blades MC, Barone F, Yanni G, Fitzgerald O, Bresnihan B, Caporali R, Montecucco C, Uguccioni M, Pitzalis C. Systematic microanatomical analysis of CXCL13 and CCL21 in situ production and progressive lymphoid organization in rheumatoid synovitis. Eur J Immunol. 2005;35:1347–1359. doi: 10.1002/eji.200425830. - DOI - PubMed
-
- Manzo A, Vitolo B, Humby F, Caporali R, Jarrossay D, Dell'accio F, Ciardelli L, Uguccioni M, Montecucco C, Pitzalis C. Mature antigen-experienced T helper cells synthesize and secrete the B cell chemoattractant CXCL13 in the inflammatory environment of the rheumatoid joint. Arthritis Rheum. 2008;58:3377–3387. doi: 10.1002/art.23966. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

