Catalyst-controlled Wacker-type oxidation of protected allylic amines
- PMID: 20799310
- PMCID: PMC3035046
- DOI: 10.1002/anie.201004156
Catalyst-controlled Wacker-type oxidation of protected allylic amines
Figures

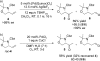
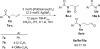
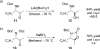
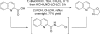
Similar articles
-
Aldehyde selective Wacker oxidations of phthalimide protected allylic amines: a new catalytic route to beta3-amino acids.J Am Chem Soc. 2009 Jul 15;131(27):9473-4. doi: 10.1021/ja902591g. J Am Chem Soc. 2009. PMID: 19583430
-
Reductive alkylation of beta-alkoxy aziridines: new route to substituted allylic amines.Org Lett. 2004 Dec 23;6(26):4817-9. doi: 10.1021/ol048201l. Org Lett. 2004. PMID: 15606074
-
A highly enantioselective allylic amination reaction using a commercially available chiral rhodium catalyst: resolution of racemic allylic carbonates.Org Lett. 2009 Jul 16;11(14):3140-2. doi: 10.1021/ol901031b. Org Lett. 2009. PMID: 19527001
-
Transition metal-catalyzed allylic substitution reactions with unactivated allylic substrates.Chem Soc Rev. 2015 Nov 21;44(22):7929-67. doi: 10.1039/c5cs00144g. Epub 2015 Aug 21. Chem Soc Rev. 2015. PMID: 26293479 Review.
-
A Regio- and Stereodivergent Synthesis of Homoallylic Amines by a One-Pot Cooperative-Catalysis-Based Allylic Alkylation/Hofmann Rearrangement Strategy.Angew Chem Int Ed Engl. 2019 Jul 29;58(31):10521-10527. doi: 10.1002/anie.201905426. Epub 2019 Jun 28. Angew Chem Int Ed Engl. 2019. PMID: 31132203 Free PMC article. Review.
Cited by
-
Mechanistic Basis for Regioselection and Regiodivergence in Nickel-Catalyzed Reductive Couplings.Acc Chem Res. 2015 Jun 16;48(6):1736-45. doi: 10.1021/acs.accounts.5b00096. Epub 2015 May 12. Acc Chem Res. 2015. PMID: 25965694 Free PMC article.
-
Imparting catalyst control upon classical palladium-catalyzed alkenyl C-H bond functionalization reactions.Acc Chem Res. 2012 Jun 19;45(6):874-84. doi: 10.1021/ar200236v. Epub 2011 Nov 23. Acc Chem Res. 2012. PMID: 22111756 Free PMC article.
-
Terminal olefins to linear α,β-unsaturated ketones: Pd(II)/hypervalent iodine co-catalyzed Wacker oxidation-dehydrogenation.J Am Chem Soc. 2013 May 29;135(21):7831-4. doi: 10.1021/ja402651q. Epub 2013 May 17. J Am Chem Soc. 2013. PMID: 23672479 Free PMC article.
-
On the mechanism of the palladium-catalyzed tert-butylhydroperoxide-mediated Wacker-type oxidation of alkenes using quinoline-2-oxazoline ligands.J Am Chem Soc. 2011 Jun 1;133(21):8317-25. doi: 10.1021/ja2017043. Epub 2011 May 9. J Am Chem Soc. 2011. PMID: 21553838 Free PMC article.
-
Catalyst-controlled Wacker-type oxidation of homoallylic alcohols in the absence of protecting groups.J Org Chem. 2011 May 6;76(9):3609-13. doi: 10.1021/jo200462a. Epub 2011 Apr 13. J Org Chem. 2011. PMID: 21446720 Free PMC article.
References
-
-
For relevant reviews see: Tsuji J. Synthesis. 1984:369–384. Takacs JM, Jiang X.-t. Curr. Org. Chem. 2003;7:369–396. Cornell CN, Sigman MS. Inorg. Chem. 2007;46:1903–1909.
-
-
-
For a review on aldehyde formation through Wacker oxidation see: Muzart J. Tetrahedron. 2007;63:7505–7521.
-
-
-
For N-based directed Wacker oxidations: Mori M, Watanabe Y, Kagechika K, Shibasaki M. Heterocycles. 1989;29:2089–2092. Hosokawa T, Aoki S, Takano M, Nakahira T, Yoshida Y, Murahashi S. J. Chem. Soc. Chem. Commun. 1991:1559–1560. Alonso RA, Burgey CS, Rao BV, Vite GD, Vollerthun R, Zottola MA, Fraser-Reid B. J. Am. Chem. Soc. 1993;115:6666–6672. Stragies R, Blechert S. J. Am. Chem. Soc. 2000;122:9584–9591. Friestad GK, Jiang T, Mathies AK. Org. Lett. 2007;9:777–780.
-
-
- Weiner B, Baeza A, Jerphagnon T, Feringa BL. J. Am. Chem. Soc. 2009;131:9473–9474. - PubMed
-
- Sigman MS, Jensen DR. Acc. Chem. Res. 2006;39:221–229. - PubMed
- Cornell CN, Sigman MS. J. Am. Chem. Soc. 2005;127:2796–2797. - PMC - PubMed
- Cornell CN, Sigman MS. Org. Lett. 2006;8:4117–4120. - PubMed
- Zhang Y, Sigman MS. J. Am. Chem. Soc. 2007;129:3076–3077. - PubMed
- Jensen KH, Pathak TP, Zhang Y, Sigman MS. J. Am. Chem. Soc. 2009;131:17074–17075. - PMC - PubMed
- Urkalan KB, Sigman MS. J. Am. Chem. Soc. 2009;131:18042–18043. - PMC - PubMed
- Werner EW, Urkalan KB, Sigman MS. Org. Lett. 2010;12:2848–2851. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

