Radiation-guided drug delivery to mouse models of lung cancer
- PMID: 20802016
- PMCID: PMC2955851
- DOI: 10.1158/1078-0432.CCR-10-0969
Radiation-guided drug delivery to mouse models of lung cancer
Abstract
Purpose: The purpose of this study was to achieve improved cancer-specific delivery and bioavailability of radiation-sensitizing chemotherapy using radiation-guided drug delivery.
Experimental design: Phage display technology was used to isolate a recombinant peptide (HVGGSSV) that binds to a radiation-inducible receptor in irradiated tumors. This peptide was used to target nab-paclitaxel to irradiated tumors, achieving tumor-specificity and enhanced bioavailability of paclitaxel.
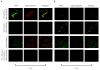
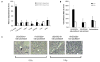
Results: Optical imaging studies showed that HVGGSSV-guided nab-paclitaxel selectively targeted irradiated tumors and showed 1.48 ± 1.66 photons/s/cm(2)/sr greater radiance compared with SGVSGHV-nab-paclitaxel, and 1.49 ± 1.36 photons/s/cm(2)/sr greater than nab-paclitaxel alone (P < 0.05). Biodistribution studies showed >5-fold increase in paclitaxel levels within irradiated tumors in HVGGSSV-nab-paclitaxel-treated groups as compared with either nab-paclitaxel or SGVSGHV-nab-paclitaxel at 72 hours. Both Lewis lung carcinoma and H460 lung carcinoma murine models showed significant tumor growth delay for HVGGSSV-nab-paclitaxel as compared with nab-paclitaxel, SGVSGHV-nab-paclitaxel,and saline controls. HVGGSSV-nab-paclitaxel treatment induced a significantly greater loss in vasculature in irradiated tumors compared with unirradiated tumors, nab-paclitaxel, SGVSGHV-nab-paclitaxel, and untreated controls.
Conclusions: HVGGSSV-nab-paclitaxel was found to bind specifically to the tax-interacting protein-1 (TIP-1) receptor expressed in irradiated tumors, enhance bioavailability of paclitaxel, and significantly increase tumor growth delay as compared with controls in mouse models of lung cancer. Here we show that targeting nab-paclitaxel to radiation-inducible TIP-1 results in increased tumor-specific drug delivery and enhanced biological efficacy in the treatment of cancer.
©2010 AACR.
Figures


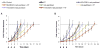
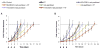
References
-
- Arap W, Pasqualini R, Ruoslahti E. Cancer treatment by targeted drug delivery to tumor vasculature in a mouse model. Science. 1998;279:377–380. - PubMed
-
- Sengupta S, Eavarone D, Capila I, et al. Temporal targeting of tumour cells and neovasculature with a nanoscale delivery system. Nature Letters. 2005;436:568–572. - PubMed
-
- Desai N, Trieu V, Yao Z, et al. Increased antitumor activity, intratumor paclitaxel concentrations, and endothelial cell transport of cremophor-free, albumin-bound paclitaxel, ABI-007, compared with Cremophor-based paclitaxel. Clinical Cancer Research. 2006;12:1317–1324. - PubMed
-
- Allen TM, Cullis PR. Drug delivery systems: entering the mainstream. Science. 2004;303:1818–1822. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

