AglJ adds the first sugar of the N-linked pentasaccharide decorating the Haloferax volcanii S-layer glycoprotein
- PMID: 20802039
- PMCID: PMC2953680
- DOI: 10.1128/JB.00705-10
AglJ adds the first sugar of the N-linked pentasaccharide decorating the Haloferax volcanii S-layer glycoprotein
Abstract
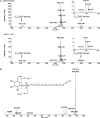
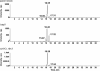
Like the Eukarya and Bacteria, the Archaea also perform N glycosylation. Using the haloarchaeon Haloferax volcanii as a model system, a series of Agl proteins involved in the archaeal version of this posttranslational modification has been identified. In the present study, the participation of HVO_1517 in N glycosylation was considered, given its homology to a known component of the eukaryal N-glycosylation pathway and because of the genomic proximity of HVO_1517 to agl genes encoding known elements of the H. volcanii N-glycosylation process. By combining the deletion of HVO_1517 with mass spectrometric analysis of both dolichol phosphate monosaccharide-charged carriers and the S-layer glycoprotein, evidence was obtained showing the participation of HVO_1517, renamed AglJ, in adding the first hexose of the N-linked pentasaccharide decorating this reporter glycoprotein. The deletion of aglJ, however, did not fully prevent the attachment of a hexose residue to the S-layer glycoprotein. Moreover, in the absence of AglJ, the level of only one of the three monosaccharide-charged dolichol phosphate carriers detected in the cell was reduced. Nonetheless, in cells lacking AglJ, no further sugar subunits were added to the remaining monosaccharide-charged dolichol phosphate carriers or to the monosaccharide-modified S-layer glycoprotein, pointing to the importance of the sugar added through the actions of AglJ for proper N glycosylation. Finally, while aglJ can be deleted, H. volcanii surface layer integrity is compromised in the absence of the encoded protein.
Figures




References
-
- Abu-Qarn, M., and J. Eichler. 2006. Protein N-glycosylation in Archaea: defining Haloferax volcanii genes involved in S-layer glycoprotein glycosylation. Mol. Microbiol. 61:511-525. - PubMed
-
- Abu-Qarn, M., S. Yurist-Doutsch, A. Giordano, A. Trauner, H. R. Morris, P. Hitchen, O. Medalia, A. Dell, and J. Eichler. 2007. Haloferax volcanii AglB and AglD are involved in N-glycosylation of the S-layer glycoprotein and proper assembly of the surface layer. J. Mol. Biol. 374:1224-1236. - PubMed
-
- Burda, P., and M. Aebi. 1999. The dolichol pathway of N-linked glycosylation. Biochim. Biophys. Acta 1426:239-257. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

