Distinct superficial and deep laminar domains of activity in the visual cortex during rest and stimulation
- PMID: 20802856
- PMCID: PMC2928665
- DOI: 10.3389/fnsys.2010.00031
Distinct superficial and deep laminar domains of activity in the visual cortex during rest and stimulation
Abstract
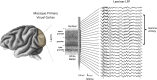
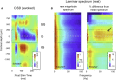
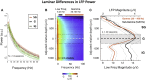
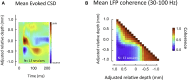
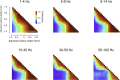
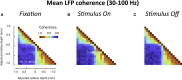
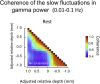
Spatial patterns of spontaneous neural activity at rest have previously been associated with specific networks in the brain, including those pertaining to the functional architecture of the primary visual cortex (V1). However, despite the prominent anatomical differences between cortical layers, little is known about the laminar pattern of spontaneous activity in V1. We address this topic by investigating the amplitude and coherence of ongoing local field potential (LFP) signals measured from different layers in V1 of macaque monkeys during rest and upon presentation of a visual stimulus. We used a linear microelectrode array to measure LFP signals at multiple, evenly spaced positions throughout the cortical thickness. Analyzing both the mean LFP amplitudes and between-contact LFP coherences, we identified two distinct zones of activity, roughly corresponding to superficial and deep layers, divided by a sharp transition near the bottom of layer 4. The LFP signals within each laminar zone were highly coherent, whereas those between zones were not. This functional compartmentalization was found not only during rest, but also when the receptive field was stimulated during a visual task. These results demonstrate the existence of distinct superficial and deep functional domains of coherent LFP activity in V1 that may reflect the intrinsic interplay of V1 microcircuitry with cortical and subcortical targets, respectively.
Keywords: LFP; V1; coherence; gamma; laminae; layers; resting state; visual perception.
Figures

References
-
- Arieli A., Shoham D., Hildesheim R., Grinvald A. (1995). Coherent spatiotemporal patterns of ongoing activity revealed by real-time optical imaging coupled with single-unit recording in the cat visual cortex. J. Neurophysiol. 73, 2072–2093 - PubMed
-
- Buffalo E. A., Fries P., Desimone R. (2004). Layer-specific attentional modulation in early visual areas. Soc. Neurosci. Abstr. Program No. 717.6
LinkOut - more resources
Full Text Sources

