TRP channels in neurogastroenterology: opportunities for therapeutic intervention
- PMID: 20804496
- PMCID: PMC3012403
- DOI: 10.1111/j.1476-5381.2010.01009.x
TRP channels in neurogastroenterology: opportunities for therapeutic intervention
Abstract
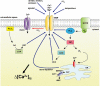
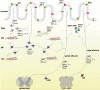
The members of the superfamily of transient receptor potential (TRP) cation channels are involved in a plethora of cellular functions. During the last decade, a vast amount of evidence is accumulating that attributes an important role to these cation channels in different regulatory aspects of the alimentary tract. In this review we discuss the expression patterns and roles of TRP channels in the regulation of gastrointestinal motility, enteric nervous system signalling and visceral sensation, and provide our perspectives on pharmacological targeting of TRPs as a strategy to treat various gastrointestinal disorders. We found that the current knowledge about the role of some members of the TRP superfamily in neurogastroenterology is rather limited, whereas the function of other TRP channels, especially of those implicated in smooth muscle cell contractility (TRPC4, TRPC6), visceral sensitivity and hypersensitivity (TRPV1, TRPV4, TRPA1), tends to be well established. Compared with expression data, mechanistic information about TRP channels in intestinal pacemaking (TRPC4, TRPC6, TRPM7), enteric nervous system signalling (TRPCs) and enteroendocrine cells (TRPM5) is lacking. It is clear that several different TRP channels play important roles in the cellular apparatus that controls gastrointestinal function. They are involved in the regulation of gastrointestinal motility and absorption, visceral sensation and visceral hypersensitivity. TRP channels can be considered as interesting targets to tackle digestive diseases, motility disorders and visceral pain. At present, TRPV1 antagonists are under development for the treatment of heartburn and visceral hypersensitivity, but interference with other TRP channels is also tempting. However, their role in gastrointestinal pathophysiology first needs to be further elucidated.
© 2010 The Authors. British Journal of Pharmacology © 2010 The British Pharmacological Society.
Figures
Similar articles
-
TRP channels in the digestive system.Curr Pharm Biotechnol. 2011 Jan 1;12(1):24-34. doi: 10.2174/138920111793937862. Curr Pharm Biotechnol. 2011. PMID: 20932260 Free PMC article. Review.
-
Transient receptor potential ion channel function in sensory transduction and cellular signaling cascades underlying visceral hypersensitivity.Am J Physiol Gastrointest Liver Physiol. 2017 Jun 1;312(6):G635-G648. doi: 10.1152/ajpgi.00401.2016. Epub 2017 Apr 6. Am J Physiol Gastrointest Liver Physiol. 2017. PMID: 28385695 Review.
-
Transient receptor potential (TRP) channels as drug targets for diseases of the digestive system.Pharmacol Ther. 2011 Jul;131(1):142-70. doi: 10.1016/j.pharmthera.2011.03.006. Epub 2011 Mar 21. Pharmacol Ther. 2011. PMID: 21420431 Free PMC article. Review.
-
An overview on transient receptor potential channels superfamily.Behav Pharmacol. 2020 Aug;31(5):413-434. doi: 10.1097/FBP.0000000000000524. Behav Pharmacol. 2020. PMID: 31833970 Review.
-
Functional expression of transient receptor potential channels in human endometrial stromal cells during the luteal phase of the menstrual cycle.Hum Reprod. 2015 Jun;30(6):1421-36. doi: 10.1093/humrep/dev068. Epub 2015 Mar 27. Hum Reprod. 2015. PMID: 25820697
Cited by
-
Irritable bowel syndrome: methods, mechanisms, and pathophysiology. Neural and neuro-immune mechanisms of visceral hypersensitivity in irritable bowel syndrome.Am J Physiol Gastrointest Liver Physiol. 2012 May 15;302(10):G1085-98. doi: 10.1152/ajpgi.00542.2011. Epub 2012 Mar 8. Am J Physiol Gastrointest Liver Physiol. 2012. PMID: 22403791 Free PMC article. Review.
-
Transient receptor potential ion channels in primary sensory neurons as targets for novel analgesics.Br J Pharmacol. 2014 May;171(10):2508-27. doi: 10.1111/bph.12532. Br J Pharmacol. 2014. PMID: 24283624 Free PMC article. Review.
-
New strategies to develop novel pain therapies: addressing thermoreceptors from different points of view.Pharmaceuticals (Basel). 2011 Dec 27;5(1):16-48. doi: 10.3390/ph5010016. Pharmaceuticals (Basel). 2011. PMID: 24288041 Free PMC article.
-
Integration of lncRNA and mRNA Transcriptome Analyses Reveals Genes and Pathways Potentially Involved in Calf Intestinal Growth and Development during the Early Weeks of Life.Genes (Basel). 2018 Mar 5;9(3):142. doi: 10.3390/genes9030142. Genes (Basel). 2018. PMID: 29510583 Free PMC article.
-
Palmitoylethanolamide normalizes intestinal motility in a model of post-inflammatory accelerated transit: involvement of CB₁ receptors and TRPV1 channels.Br J Pharmacol. 2014 Sep;171(17):4026-37. doi: 10.1111/bph.12759. Br J Pharmacol. 2014. PMID: 24818658 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

