A testis-specific regulator of complex and hybrid N-glycan synthesis
- PMID: 20805325
- PMCID: PMC2935569
- DOI: 10.1083/jcb.201004102
A testis-specific regulator of complex and hybrid N-glycan synthesis
Abstract
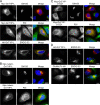
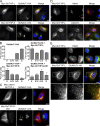
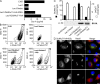
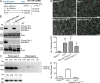
Database analyses identified 4933434I20Rik as a glycosyltransferase-like gene expressed mainly in testicular germ cells and regulated during spermatogenesis. Expression of a membrane-bound form of the protein resulted in a marked and specific reduction in N-acetylglucosaminyltransferase I (GlcNAcT-I) activity and complex and hybrid N-glycan synthesis. Thus, the novel activity was termed GlcNAcT-I inhibitory protein (GnT1IP). Membrane-bound GnT1IP localizes to the ER, the ER-Golgi intermediate compartment (ERGIC), and the cis-Golgi. Coexpression of membrane-anchored GnT1IP with GlcNAcT-I causes association of the two proteins, inactivation of GlcNAcT-I, and mislocalization of GlcNAcT-I from the medial-Golgi to earlier compartments. Therefore, GnT1IP is a regulator of GlcNAcT-I and complex and hybrid N-glycan production. Importantly, the formation of high mannose N-glycans resulting from inhibition of GlcNAcT-I by GnT1IP markedly increases the adhesion of CHO cells to TM4 Sertoli cells. Testicular germ cells might use GnT1IP to induce the expression of high mannose N-glycans on glycoproteins, thereby facilitating Sertoli-germ cell attachment at a particular stage of spermatogenesis.
Figures





References
-
- Akama T.O., Nakagawa H., Wong N.K., Sutton-Smith M., Dell A., Morris H.R., Nakayama J., Nishimura S., Pai A., Moremen K.W., et al. 2006. Essential and mutually compensatory roles of alpha-mannosidase II and alpha-mannosidase IIx in N-glycan processing in vivo in mice. Proc. Natl. Acad. Sci. USA. 103:8983–8988. 10.1073/pnas.0603248103 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

