Arabidopsis basic leucine-zipper transcription factors TGA9 and TGA10 interact with floral glutaredoxins ROXY1 and ROXY2 and are redundantly required for anther development
- PMID: 20805327
- PMCID: PMC2971623
- DOI: 10.1104/pp.110.159111
Arabidopsis basic leucine-zipper transcription factors TGA9 and TGA10 interact with floral glutaredoxins ROXY1 and ROXY2 and are redundantly required for anther development
Erratum in
-
CORRECTION: Vol. 154: 1492-1504, 2010.Plant Physiol. 2018 Jul;177(3):1342-1343. doi: 10.1104/pp.18.00667. Epub 2018 May 30. Plant Physiol. 2018. PMID: 29848747 Free PMC article. No abstract available.
Abstract
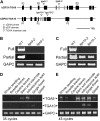
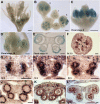
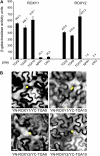
ROXY1 and ROXY2 are CC-type floral glutaredoxins with redundant functions in Arabidopsis (Arabidopsis thaliana) anther development. We show here that plants lacking the basic leucine-zipper transcription factors TGA9 and TGA10 have defects in male gametogenesis that are strikingly similar to those in roxy1 roxy2 mutants. In tga9 tga10 mutants, adaxial and abaxial anther lobe development is differentially affected, with early steps in anther development blocked in adaxial lobes and later steps affected in abaxial lobes. Distinct from roxy1 roxy2, microspore development in abaxial anther lobes proceeds to a later stage with the production of inviable pollen grains contained within nondehiscent anthers. Histological analysis shows multiple defects in the anther dehiscence program, including abnormal stability and lignification of the middle layer and defects in septum and stomium function. Compatible with these defects, TGA9 and TGA10 are expressed throughout early anther primordia but resolve to the middle and tapetum layers during meiosis of pollen mother cells. Several lines of evidence suggest that ROXY promotion of anther development is mediated in part by TGA9 and TGA10. First, TGA9 and TGA10 expression overlaps with ROXY1/2 during anther development. Second, TGA9/10 and ROXY1/2 operate downstream of SPOROCYTELESS/NOZZLE, where they positively regulate a common set of genes that contribute to tapetal development. Third, TGA9 and TGA10 directly interact with ROXY proteins in yeast and in plant cell nuclei. These findings suggest that activation of TGA9/10 transcription factors by ROXY-mediated modification of cysteine residues promotes anther development, thus broadening our understanding of how redox-regulated TGA factors function in plants.
Figures




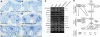
Similar articles
-
ROXY1 and ROXY2, two Arabidopsis glutaredoxin genes, are required for anther development.Plant J. 2008 Mar;53(5):790-801. doi: 10.1111/j.1365-313X.2007.03375.x. Epub 2007 Nov 23. Plant J. 2008. PMID: 18036205
-
Repression of the Arabidopsis thaliana jasmonic acid/ethylene-induced defense pathway by TGA-interacting glutaredoxins depends on their C-terminal ALWL motif.Mol Plant. 2012 Jul;5(4):831-40. doi: 10.1093/mp/ssr113. Epub 2011 Dec 29. Mol Plant. 2012. PMID: 22207719
-
Arabidopsis clade IV TGA transcription factors, TGA10 and TGA9, are involved in ROS-mediated responses to bacterial PAMP flg22.Plant Sci. 2016 Nov;252:12-21. doi: 10.1016/j.plantsci.2016.06.019. Epub 2016 Jun 28. Plant Sci. 2016. PMID: 27717447
-
New players unveiled in early anther development.Plant Signal Behav. 2011 Jul;6(7):934-8. doi: 10.4161/psb.6.7.15668. Plant Signal Behav. 2011. PMID: 21633200 Free PMC article. Review.
-
The final split: the regulation of anther dehiscence.J Exp Bot. 2011 Mar;62(5):1633-49. doi: 10.1093/jxb/err014. Epub 2011 Feb 16. J Exp Bot. 2011. PMID: 21325605 Review.
Cited by
-
Genome-Wide Identification of the TGA Gene Family and Expression Analysis under Drought Stress in Brassica napus L.Int J Mol Sci. 2024 Jun 8;25(12):6355. doi: 10.3390/ijms25126355. Int J Mol Sci. 2024. PMID: 38928064 Free PMC article.
-
Unveiling the Redox Control of Plant Reproductive Development during Abiotic Stress.Front Plant Sci. 2016 Jun 16;7:700. doi: 10.3389/fpls.2016.00700. eCollection 2016. Front Plant Sci. 2016. PMID: 27379102 Free PMC article. Review.
-
PsEND1 Is a Key Player in Pea Pollen Development Through the Modulation of Redox Homeostasis.Front Plant Sci. 2021 Oct 29;12:765277. doi: 10.3389/fpls.2021.765277. eCollection 2021. Front Plant Sci. 2021. PMID: 34777450 Free PMC article.
-
Genome-Wide Characterization and Expression Analysis of Soybean TGA Transcription Factors Identified a Novel TGA Gene Involved in Drought and Salt Tolerance.Front Plant Sci. 2019 May 16;10:549. doi: 10.3389/fpls.2019.00549. eCollection 2019. Front Plant Sci. 2019. PMID: 31156656 Free PMC article.
-
Glutaredoxins are essential for stress adaptation in the cyanobacterium Synechocystis sp. PCC 6803.Front Plant Sci. 2013 Nov 4;4:428. doi: 10.3389/fpls.2013.00428. eCollection 2013. Front Plant Sci. 2013. PMID: 24204369 Free PMC article.
References
-
- Aarts MGM, Hodge R, Kalantidis K, Florack D, Wilson ZA, Mulligan BJ, Stiekema WJ, Scott R, Pereira A. (1997) The Arabidopsis MALE STERILITY 2 protein shares similarity with reductases in elongation/condensation complexes. Plant J 12: 615–623 - PubMed
-
- Alexander MP. (1969) Differential staining of aborted and nonaborted pollen. Stain Technol 44: 117–122 - PubMed
-
- Alvarez S, Zhu M, Chen S. (2009) Proteomics of Arabidopsis redox proteins in response to methyl jasmonate. J Proteomics 73: 30–40 - PubMed
-
- Apel K, Hirt H. (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55: 373–399 - PubMed
-
- Ariizumi T, Hatakeyama K, Hinata K, Inatsugi R, Nishida I, Sato S, Kato T, Tabata S, Toriyama K. (2004) Disruption of the novel plant protein NEF1 affects lipid accumulation in the plastids of the tapetum and exine formation of pollen, resulting in male sterility in Arabidopsis thaliana. Plant J 39: 170–181 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

