Standards for immunohistochemical imaging: a protein reference device for biomarker quantitation
- PMID: 20805583
- PMCID: PMC2958133
- DOI: 10.1369/jhc.2010.956342
Standards for immunohistochemical imaging: a protein reference device for biomarker quantitation
Abstract
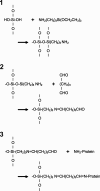
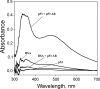
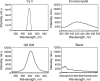
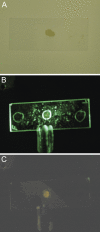
We are developing a reference device to be used in the validation of immunohistochemical imaging of biomarkers by microscopy. The prototype device consists of p53 protein immobilized at various concentrations on a glass slide. The device is designed as a reference control to be used with assays that incorporate commercially available anti-p53 antibodies. p53 protein was characterized by mass spectrometry and covalently immobilized through amide linkage to the (3-aminopropyl)trietoxysilane-modified glass surface. This procedure is reproducible and provides a chemically stable product in high yield. The surface-bound protein was shown to be immunoreactive by its specific interaction with anti-p53 antibody (Ab) and detection by absorbance and fluorescence spectroscopy. Also, comparison was made with microscopic images of Ab-stained tissue samples, known to stain positive for p53. Further development will be required to establish accurate surface protein concentrations in the range required for specific clinical applications.
Figures


Similar articles
-
Interaction of an anticancer peptide fragment of azurin with p53 and its isolated domains studied by atomic force spectroscopy.Int J Nanomedicine. 2011;6:3011-9. doi: 10.2147/IJN.S26155. Epub 2011 Nov 24. Int J Nanomedicine. 2011. PMID: 22162658 Free PMC article.
-
Toward Liquid Biopsy: Determination of the Humoral Immune Response in Cancer Patients Using HaloTag Fusion Protein-Modified Electrochemical Bioplatforms.Anal Chem. 2016 Dec 20;88(24):12339-12345. doi: 10.1021/acs.analchem.6b03526. Epub 2016 Dec 1. Anal Chem. 2016. PMID: 28193070
-
Spectroscopic and microscopic characterization of biosensor surfaces with protein/amino-organosilane/silicon structure.Colloids Surf B Biointerfaces. 2012 Feb 1;90:159-68. doi: 10.1016/j.colsurfb.2011.10.017. Epub 2011 Oct 18. Colloids Surf B Biointerfaces. 2012. PMID: 22056253
-
On typing amyloidosis using immunohistochemistry. Detailled illustrations, review and a note on mass spectrometry.Prog Histochem Cytochem. 2012 Aug;47(2):61-132. doi: 10.1016/j.proghi.2012.03.001. Epub 2012 Jul 20. Prog Histochem Cytochem. 2012. PMID: 22819384 Review.
-
Garbage in, garbage out: a critical evaluation of strategies used for validation of immunohistochemical biomarkers.Mol Oncol. 2014 Jun;8(4):783-98. doi: 10.1016/j.molonc.2014.03.008. Epub 2014 Mar 21. Mol Oncol. 2014. PMID: 24725481 Free PMC article. Review.
Cited by
-
Biomolecular Interactions of Cytotoxic Ruthenium Compounds with Thiosemicarbazone or Benzothiazole Schiff Base Chelates.ChemMedChem. 2022 Oct 19;17(20):e202200444. doi: 10.1002/cmdc.202200444. Epub 2022 Sep 21. ChemMedChem. 2022. PMID: 36041073 Free PMC article.
-
High throughput screening of mesenchymal stem cell lines using deep learning.Sci Rep. 2022 Oct 20;12(1):17507. doi: 10.1038/s41598-022-21653-y. Sci Rep. 2022. PMID: 36266301 Free PMC article.
-
Companion diagnostic requirements for spatial biology using multiplex immunofluorescence and multispectral imaging.Front Mol Biosci. 2023 Feb 9;10:1051491. doi: 10.3389/fmolb.2023.1051491. eCollection 2023. Front Mol Biosci. 2023. PMID: 36845550 Free PMC article. Review.
-
Synthetic Antigen Gels as Practical Controls for Standardized and Quantitative Immunohistochemistry.J Histochem Cytochem. 2019 May;67(5):309-334. doi: 10.1369/0022155419832002. Epub 2019 Mar 18. J Histochem Cytochem. 2019. PMID: 30879407 Free PMC article.
References
-
- Atha DH, Miller K, Sanow AD, Xu J, Hess JL, Wu OC, Wang W, et al. (2003) High-throughput analysis of telomerase by capillary electrophoresis. Electrophoresis 24:109–114 - PubMed
-
- Atha DH, Wenz H-M, Morehead H, Tian J, O'Connell C (1998) Detection of p53 point mutations by single strand conformation polymorphism: analysis by capillary electrophoresis. Electrophoresis 19:172–179 - PubMed
-
- Barker PE (2003) Cancer biomarker validation: standards and process: roles for the National Institute of Standards and Technology (NIST). Ann NY Acad Sci 983:142–150 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

