Caylobolide B, a macrolactone from symplostatin 1-producing marine cyanobacteria Phormidium spp. from Florida
- PMID: 20806908
- PMCID: PMC2965599
- DOI: 10.1021/np100467d
Caylobolide B, a macrolactone from symplostatin 1-producing marine cyanobacteria Phormidium spp. from Florida
Erratum in
-
Correction to Caylobolide B, a Macrolactone from Symplostatin 1-Producing Marine Cyanobacteria Phormidium spp. from Florida.J Nat Prod. 2016 Feb 26;79(2):452. doi: 10.1021/acs.jnatprod.6b00120. Epub 2016 Feb 17. J Nat Prod. 2016. PMID: 26885760 No abstract available.
Abstract
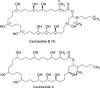
A Phormidium spp. collection from Key West, Florida, afforded caylobolide B (1), an analogue of the known macrolactone caylobolide A, previously isolated from a Lyngbya majuscula collection from the Bahamas. The planar structure of 1 was determined using NMR and MS experiments. The relative configuration for subunits C7-C9 and C25-C29 was assigned using Kishi's Universal NMR Database. Caylobolide B (1) displayed cytotoxic activity against HT29 colorectal adenocarcinoma and HeLa cervical carcinoma cells with IC(50) values of 4.5 and 12.2 μM, respectively.
Figures

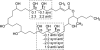
Similar articles
-
Caylobolide A, a unique 36-membered macrolactone from a Bahamian Lyngbya majuscula.Org Lett. 2002 May 2;4(9):1535-8. doi: 10.1021/ol025759p. Org Lett. 2002. PMID: 11975622
-
Hantupeptin A, a cytotoxic cyclic depsipeptide from a Singapore collection of Lyngbya majuscula.J Nat Prod. 2009 Jan;72(1):29-32. doi: 10.1021/np800448t. J Nat Prod. 2009. PMID: 19093843
-
Malyngamide 3 and cocosamides A and B from the marine cyanobacterium Lyngbya majuscula from Cocos Lagoon, Guam.J Nat Prod. 2011 Apr 25;74(4):871-6. doi: 10.1021/np1008015. Epub 2011 Feb 22. J Nat Prod. 2011. PMID: 21341718 Free PMC article.
-
Isolation and biological evaluation of 8-epi-malyngamide C from the Floridian marine cyanobacterium Lyngbya majuscula.J Nat Prod. 2010 Mar 26;73(3):463-6. doi: 10.1021/np900614n. J Nat Prod. 2010. PMID: 20166701 Free PMC article.
-
Total structure determination of grassypeptolide, a new marine cyanobacterial cytotoxin.Org Lett. 2008 Mar 6;10(5):789-92. doi: 10.1021/ol702946d. Epub 2008 Jan 26. Org Lett. 2008. PMID: 18220404
Cited by
-
Nanosynthesis, phycochemical constituents, and pharmacological properties of cyanobacterium Oscillatoria sp.Naunyn Schmiedebergs Arch Pharmacol. 2024 Mar;397(3):1347-1375. doi: 10.1007/s00210-023-02719-8. Epub 2023 Sep 15. Naunyn Schmiedebergs Arch Pharmacol. 2024. PMID: 37712972 Review.
-
Bioprospecting Sponge-Associated Marine Cyanobacteria to Produce Bioactive Compounds.Toxins (Basel). 2020 Jan 23;12(2):73. doi: 10.3390/toxins12020073. Toxins (Basel). 2020. PMID: 31979262 Free PMC article.
-
Chemical diversity of cyanobacterial natural products.Nat Prod Rep. 2025 Jan 22;42(1):6-49. doi: 10.1039/d4np00040d. Nat Prod Rep. 2025. PMID: 39540765 Review.
-
Marine Cyanobacteria as Sources of Lead Anticancer Compounds: A Review of Families of Metabolites with Cytotoxic, Antiproliferative, and Antineoplastic Effects.Molecules. 2022 Jul 27;27(15):4814. doi: 10.3390/molecules27154814. Molecules. 2022. PMID: 35956762 Free PMC article. Review.
-
Marine Cyanobacteria and Microalgae Metabolites-A Rich Source of Potential Anticancer Drugs.Mar Drugs. 2020 Sep 19;18(9):476. doi: 10.3390/md18090476. Mar Drugs. 2020. PMID: 32961827 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

