Randomized placebo-controlled trial of prednisone for paradoxical tuberculosis-associated immune reconstitution inflammatory syndrome
- PMID: 20808204
- PMCID: PMC2940061
- DOI: 10.1097/QAD.0b013e32833dfc68
Randomized placebo-controlled trial of prednisone for paradoxical tuberculosis-associated immune reconstitution inflammatory syndrome
Abstract
Objective: Paradoxical tuberculosis-associated immune reconstitution inflammatory syndrome (TB-IRIS) is a frequent complication of antiretroviral therapy in resource-limited countries. We aimed to assess whether a 4-week course of prednisone would reduce morbidity in patients with paradoxical TB-IRIS without excess adverse events.
Design: A randomized, double-blind, placebo-controlled trial of prednisone (1.5 mg/kg per day for 2 weeks then 0.75 mg/kg per day for 2 weeks). Patients with immediately life-threatening TB-IRIS manifestations were excluded.
Methods: The primary combined endpoint was days of hospitalization and outpatient therapeutic procedures, which were counted as one hospital day.
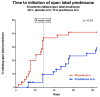
Results: One hundred and ten participants were enrolled (55 to each arm). The primary combined endpoint was more frequent in the placebo than the prednisone arm {median hospital days 3 [interquartile range (IQR) 0-9] and 0 (IQR 0-3), respectively; P = 0.04}. There were significantly greater improvements in symptoms, Karnofsky score, and quality of life (MOS-HIV) in the prednisone vs. the placebo arm at 2 and 4 weeks, but not at later time points. Chest radiographs improved significantly more in the prednisone arm at weeks 2 (P = 0.002) and 4 (P = 0.02). Infections on study medication occurred in more participants in prednisone than in placebo arm (27 vs. 17, respectively; P = 0.05), but there was no difference in severe infections (2 vs. 4, respectively; P = 0.40). Isolates from 10 participants were found to be resistant to rifampicin after enrolment.
Conclusion: Prednisone reduced the need for hospitalization and therapeutic procedures and hastened improvements in symptoms, performance, and quality of life. It is important to investigate for drug-resistant tuberculosis and other causes for deterioration before administering glucocorticoids.
Conflict of interest statement
Figures



References
-
- McIlleron H, Meintjes G, Burman WJ, Maartens G. Complications of antiretroviral therapy in patients with tuberculosis: drug interactions, toxicity, and immune reconstitution inflammatory syndrome. J Infect Dis. 2007;196(Suppl 1):S63–75. - PubMed
-
- Lawn SD, Myer L, Bekker LG, Wood R. Tuberculosis-associated immune reconstitution disease: incidence, risk factors and impact in an antiretroviral treatment service in South Africa. AIDS. 2007;21:335–341. - PubMed
-
- Bourgarit A, Carcelain G, Martinez V, Lascoux C, Delcey V, Gicquel B, et al. Explosion of tuberculin-specific Th1-responses induces immune restoration syndrome in tuberculosis and HIV co-infected patients. Aids. 2006;20:F1–7. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

