Adenovirus-retrovirus hybrid vectors achieve highly enhanced tumor transduction and antitumor efficacy in vivo
- PMID: 20808291
- PMCID: PMC3017434
- DOI: 10.1038/mt.2010.182
Adenovirus-retrovirus hybrid vectors achieve highly enhanced tumor transduction and antitumor efficacy in vivo
Abstract
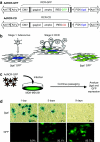
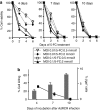
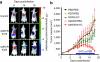
Murine leukemia virus (MLV)-based replication-competent retrovirus (RCR) vectors have been shown to mediate efficient, selective, and persistent tumor transduction, thereby achieving significant therapeutic benefit in a wide variety of cancer models. To further augment the efficiency of this strategy, we have developed a delivery method employing a gutted adenovirus encoding an RCR vector (AdRCR); thus, tumor cells transduced with the adenoviral vector transiently become RCR vector producer cells in situ. As expected, high-titer AdRCR achieved significantly higher initial transduction levels in human cancer cells both in vitro and in vivo, as compared to the original RCR vector itself. Notably, even at equivalent initial transduction levels, more secondary RCR progeny were produced from AdRCR-transduced cells as compared to RCR-transduced cells, resulting in further acceleration of subsequent RCR replication kinetics. In pre-established tumor models in vivo, prodrug activator gene therapy with high-titer AdRCR could achieve enhanced efficacy compared to RCR alone, in a dose-dependent manner. Thus, AdRCR hybrid vectors offer the advantages of high production titers characteristic of adenovirus and secondary production of RCR in situ, which not only accelerates subsequent vector spread and progressive tumor transduction, but can also significantly enhance the therapeutic efficacy of RCR-mediated prodrug activator gene therapy.
Figures


Similar articles
-
Replication-competent retrovirus vector-mediated prodrug activator gene therapy in experimental models of human malignant mesothelioma.Cancer Gene Ther. 2011 Aug;18(8):571-8. doi: 10.1038/cgt.2011.25. Epub 2011 Jun 10. Cancer Gene Ther. 2011. PMID: 21660062 Free PMC article.
-
Beyond oncolytic virotherapy: replication-competent retrovirus vectors for selective and stable transduction of tumors.Curr Gene Ther. 2005 Dec;5(6):655-67. doi: 10.2174/156652305774964659. Curr Gene Ther. 2005. PMID: 16457654 Review.
-
VSV-G pseudotyped, MuLV-based, semi-replication-competent retrovirus for cancer treatment.Gene Ther. 2006 Oct;13(20):1457-70. doi: 10.1038/sj.gt.3302782. Epub 2006 May 25. Gene Ther. 2006. PMID: 16724095
-
Use of replication-competent retroviral vectors in an immunocompetent intracranial glioma model.Neurosurg Focus. 2006 Apr 15;20(4):E25. doi: 10.3171/foc.2006.20.4.1. Neurosurg Focus. 2006. PMID: 16709031 Free PMC article.
-
Replication-competent retrovirus vectors for cancer gene therapy.Front Biosci. 2008 Jan 1;13:3083-95. doi: 10.2741/2910. Front Biosci. 2008. PMID: 17981778 Review.
Cited by
-
Highly efficient tumor transduction and antitumor efficacy in experimental human malignant mesothelioma using replicating gibbon ape leukemia virus.Cancer Gene Ther. 2013 Dec;20(12):671-7. doi: 10.1038/cgt.2013.67. Epub 2013 Nov 8. Cancer Gene Ther. 2013. PMID: 24201868 Free PMC article.
-
Efficient tumor transduction and antitumor efficacy in experimental human osteosarcoma using retroviral replicating vectors.Cancer Gene Ther. 2019 Feb;26(1-2):41-47. doi: 10.1038/s41417-018-0037-y. Epub 2018 Jul 25. Cancer Gene Ther. 2019. PMID: 30042500 Free PMC article.
-
Oncolytic Viruses-Interaction of Virus and Tumor Cells in the Battle to Eliminate Cancer.Front Oncol. 2017 Sep 8;7:195. doi: 10.3389/fonc.2017.00195. eCollection 2017. Front Oncol. 2017. PMID: 28944214 Free PMC article. Review.
-
Adenovirus vectors in hematopoietic stem cell genome editing.FEBS Lett. 2019 Dec;593(24):3623-3648. doi: 10.1002/1873-3468.13668. Epub 2019 Nov 20. FEBS Lett. 2019. PMID: 31705806 Free PMC article. Review.
-
Integration profile and safety of an adenovirus hybrid-vector utilizing hyperactive sleeping beauty transposase for somatic integration.PLoS One. 2013 Oct 4;8(10):e75344. doi: 10.1371/journal.pone.0075344. eCollection 2013. PLoS One. 2013. PMID: 24124483 Free PMC article.
References
-
- Liu TC., and, Kirn D. Gene therapy progress and prospects cancer: oncolytic viruses. Gene Ther. 2008;15:877–884. - PubMed
-
- Logg CR., and, Kasahara N. Retrovirus-mediated gene transfer to tumors: utilizing the replicative power of viruses to achieve highly efficient tumor transduction in vivo. Methods Mol Biol. 2004;246:499–525. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

