Polyphosphate kinase 2: a novel determinant of stress responses and pathogenesis in Campylobacter jejuni
- PMID: 20808906
- PMCID: PMC2923150
- DOI: 10.1371/journal.pone.0012142
Polyphosphate kinase 2: a novel determinant of stress responses and pathogenesis in Campylobacter jejuni
Abstract
Background: Inorganic polyphosphate (poly P) plays an important role in stress tolerance and virulence in many bacteria. PPK1 is the principal enzyme involved in poly P synthesis, while PPK2 uses poly P to generate GTP, a signaling molecule that serves as an alternative energy source and a precursor for various physiological processes. Campylobacter jejuni, an important cause of foodborne gastroenteritis in humans, possesses homologs of both ppk1 and ppk2. ppk1 has been previously shown to impact the pathobiology of C. jejuni.
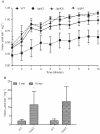
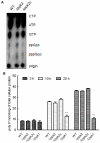
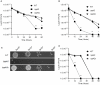
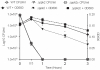
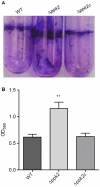
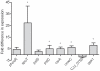
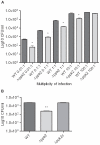
Methodology/principal findings: Here, we demonstrate for the first time that the deletion of ppk2 in C. jejuni resulted in a significant decrease in poly P-dependent GTP synthesis, while displaying an increased intracellular ATP:GTP ratio. The Deltappk2 mutant exhibited a significant survival defect under osmotic, nutrient, aerobic, and antimicrobial stresses and displayed an enhanced ability to form static biofilms. However, the Deltappk2 mutant was not defective in poly P and ppGpp synthesis suggesting that PPK2-mediated stress tolerance is not ppGpp-mediated. Importantly, the Deltappk2 mutant was significantly attenuated in invasion and intracellular survival within human intestinal epithelial cells as well as in chicken colonization.
Conclusions/significance: Taken together, we have highlighted the role of PPK2 as a novel pathogenicity determinant that is critical for C. jejuni survival, adaptation, and persistence in the host environments. PPK2 is absent in humans and animals; therefore, can serve as a novel target for therapeutic intervention of C. jejuni infections.
Conflict of interest statement
Figures

References
-
- Allos BM. Campylobacter jejuni infections: update on emerging issues and trends. Clin Infect Dis. 2001;32:1201–1206. - PubMed
-
- Hughes R. Campylobacter jejuni in Guillain-Barré syndrome. Lancet Neurol. 2004;3:644. - PubMed
-
- Kalischuk LD, Buret AG. A role for Campylobacter jejuni-induced enteritis in inflammatory bowel disease? Am J Physiol Gastrointest Liver Physiol. 2010;298:G1–G9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

