bFGF regulates PI3-kinase-Rac1-JNK pathway and promotes fibroblast migration in wound healing
- PMID: 20808927
- PMCID: PMC2923192
- DOI: 10.1371/journal.pone.0012228
bFGF regulates PI3-kinase-Rac1-JNK pathway and promotes fibroblast migration in wound healing
Abstract
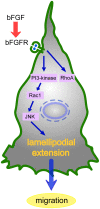
Fibroblast proliferation and migration play important roles in wound healing. bFGF is known to promote both fibroblast proliferation and migration during the process of wound healing. However, the signal transduction of bFGF-induced fibroblast migration is still unclear, because bFGF can affect both proliferation and migration. Herein, we investigated the effect of bFGF on fibroblast migration regardless of its effect on fibroblast proliferation. We noticed involvement of the small GTPases of the Rho family, PI3-kinase, and JNK. bFGF activated RhoA, Rac1, PI3-kinase, and JNK in cultured fibroblasts. Inhibition of RhoA did not block bFGF-induced fibroblast migration, whereas inhibition of Rac1, PI3-kinase, or JNK blocked the fibroblast migration significantly. PI3-kinase-inhibited cells down-regulated the activities of Rac1 and JNK, and Rac1-inhibited cells down-regulated JNK activity, suggesting that PI3-kinase is upstream of Rac1 and that JNK is downstream of Rac1. Thus, we concluded that PI3-kinase, Rac1, and JNK were essential for bFGF-induced fibroblast migration, which is a novel pathway of bFGF-induced cell migration.
Conflict of interest statement
Figures

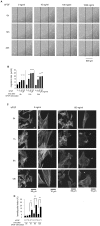

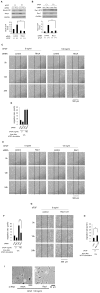
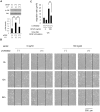
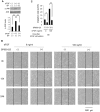

References
-
- Lauffenburger DA, Horwitz AF. Cell migration: a physically integrated molecular process. Cell. 1996;84:359–369. - PubMed
-
- Webb DJ, Parsons JT, Horwitz AF. Adhesion assembly, disassembly and turnover in migrating cells – over and over and over again. Nat Cell Biol. 2002;4:E97–E100. - PubMed
-
- Ridley AJ, Schwartz MA, Burridge K, Firtel RA, Ginsberg MH, et al. Cell migration: integrating signals from front to back. Science. 2003;302:1704–1709. - PubMed
-
- Smith LG, Li R. Actin polymerization: riding the wave. Curr Biol. 2004;14:R109–111. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

