Nogo receptor deletion and multimodal exercise improve distinct aspects of recovery in cervical spinal cord injury
- PMID: 20809785
- PMCID: PMC2978056
- DOI: 10.1089/neu.2010.1491
Nogo receptor deletion and multimodal exercise improve distinct aspects of recovery in cervical spinal cord injury
Abstract
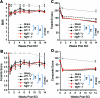
We tested the ability of two plasticity-promoting approaches to enhance recovery in a mouse model of incomplete spinal cord injury (SCI). Genetically, we reduced myelin-mediated inhibition of neural plasticity through Nogo66-receptor (NgR) gene deletion. Behaviorally, we utilized a novel multimodal exercise training paradigm. Adult mice of wild-type or NgR-null genotype were subjected to partial lateral hemisection (LHx) at C3-C4 with the intent of producing anatomically and functionally mild deficits. Exercise training or control treatment proceeded for 14 weeks. Behavioral outcomes were assessed prior to tract tracing and histological analysis. Genotype and training exerted differing effects on performance; training improved performance on a test related to the training regimen (task-specific benefit), whereas genotype also improved performance on more generalized behaviors (task-non-specific benefit). There were no significant histological differences across genotype or training assignment with regard to lesion size or axonal tract staining. Thus either NgR gene deletion or exercise training benefits mice with mild cervical spinal injury. In this lesion model, the effects of NgR deletion and training were not synergistic for the tasks assessed. Further work is required to optimize the interaction between pharmacological and physical interventions for SCI.
Figures




References
-
- Ballermann M. Fouad K. Spontaneous locomotor recovery in spinal cord injured rats is accompanied by anatomical plasticity of reticulospinal fibers. Eur. J. Neurosci. 2006;23:1988–1996. - PubMed
-
- Barbeau H. Rossignol S. Recovery of locomotion after chronic spinalization in the adult cat. Brain Res. 1987;412:84–95. - PubMed
-
- Bareyre F.M. Kerschensteiner M. Raineteau O. Mettenleiter T.C. Weinmann O. Schwab M.E. The injured spinal cord spontaneously forms a new intraspinal circuit in adult rats. Nat. Neurosci. 2004;7:269–277. - PubMed
-
- Basso D.M. Fisher L.C. Anderson A.J. Jakeman L.B. McTigue D.M. Popovich P.G. Basso Mouse Scale for locomotion detects differences in recovery after spinal cord injury in five common mouse strains. J. Neurotrauma. 2006;23:635–659. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

