CXCL10 is required to maintain T-cell populations and to control parasite replication during chronic ocular toxoplasmosis
- PMID: 20811054
- PMCID: PMC3053287
- DOI: 10.1167/iovs.10-5819
CXCL10 is required to maintain T-cell populations and to control parasite replication during chronic ocular toxoplasmosis
Abstract
Purpose: Toxoplasma gondii is a major cause of ocular disease, which can lead to permanent vision loss in humans. T cells are critically involved in parasite control, but little is known about the molecules that promote T-cell trafficking and migration in the retina. Thus, the aim of this study was to image and dissect the T-cell response during chronic toxoplasmic retinochoroiditis.
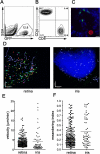
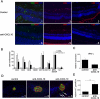
Methods: C57BL/6 mice were infected with the Me49 strain of T. gondii, and T cells that infiltrated the eye were analyzed by flow cytometry and imaged using multiphoton microscopy. IFN-γ, CXCL9, CXCL10, and CXCR3 mRNA levels were measured by real-time PCR. To investigate the role of CXCL10, mice were treated with anti-CXCL10 antibodies, and histopathology and immunohistochemistry were performed to monitor changes in pathology, cellular infiltration, and parasite burden in the eye.
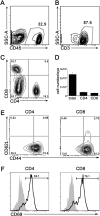
Results: Infection with T. gondii leads to the infiltration of highly activated motile T cells into the eye. These cells express CXCR3 and are capable of producing IFN-γ and TNF-α, and CD8+ T cells express granzyme B. The expression of CXCL9 and CXCL10 in the retina was significantly upregulated during chronic infection. Treatment of chronically infected mice with anti-CXCL10 antibodies led to decreases in the numbers of CD3+, CD4+, and CD8+ T cells and the amount of IFN-γ mRNA expression in the retina and an increase in replicating parasites and ocular pathology.
Conclusions: The maintenance of the T-cell response and the control of T. gondii in the eye during chronic infection is dependent on CXCL10.
Figures



Similar articles
-
Predominant interferon-γ-mediated expression of CXCL9, CXCL10, and CCL5 proteins in the brain during chronic infection with Toxoplasma gondii in BALB/c mice resistant to development of toxoplasmic encephalitis.J Interferon Cytokine Res. 2010 Sep;30(9):653-60. doi: 10.1089/jir.2009.0119. J Interferon Cytokine Res. 2010. PMID: 20626297 Free PMC article.
-
NOD2 regulates CXCR3-dependent CD8+ T cell accumulation in intestinal tissues with acute injury.J Immunol. 2014 Apr 1;192(7):3409-18. doi: 10.4049/jimmunol.1302436. Epub 2014 Mar 3. J Immunol. 2014. PMID: 24591373 Free PMC article.
-
CXC chemokine ligand (CXCL) 9 and CXCL10 are antagonistic costimulation molecules during the priming of alloreactive T cell effectors.J Immunol. 2010 Apr 1;184(7):3450-60. doi: 10.4049/jimmunol.0903831. Epub 2010 Mar 1. J Immunol. 2010. PMID: 20194716 Free PMC article.
-
The causes of tissue damage in toxoplasmic retinochoroiditis.Trans Ophthalmol Soc U K (1962). 1986;105 ( Pt 4):404-12. Trans Ophthalmol Soc U K (1962). 1986. PMID: 3538518 Review.
-
Pathogenesis of toxoplasmic retinochoroiditis.Parasitol Today. 1999 Feb;15(2):51-7. doi: 10.1016/s0169-4758(98)01377-5. Parasitol Today. 1999. PMID: 10234186 Review.
Cited by
-
Dealing with Danger in the CNS: The Response of the Immune System to Injury.Neuron. 2015 Jul 1;87(1):47-62. doi: 10.1016/j.neuron.2015.05.019. Neuron. 2015. PMID: 26139369 Free PMC article. Review.
-
Overproduction of Toxoplasma gondii cyclophilin-18 regulates host cell migration and enhances parasite dissemination in a CCR5-independent manner.BMC Microbiol. 2014 Mar 25;14:76. doi: 10.1186/1471-2180-14-76. BMC Microbiol. 2014. PMID: 24661782 Free PMC article.
-
Human Retinal Organoid Model of Ocular Toxoplasmosis.Pathogens. 2025 Mar 14;14(3):286. doi: 10.3390/pathogens14030286. Pathogens. 2025. PMID: 40137771 Free PMC article.
-
Chitinase dependent control of protozoan cyst burden in the brain.PLoS Pathog. 2012;8(11):e1002990. doi: 10.1371/journal.ppat.1002990. Epub 2012 Nov 29. PLoS Pathog. 2012. PMID: 23209401 Free PMC article.
-
Identification of a novel neurotrophic factor from primary retinal Müller cells using stable isotope labeling by amino acids in cell culture (SILAC).Mol Cell Proteomics. 2014 Sep;13(9):2371-81. doi: 10.1074/mcp.M113.033613. Epub 2014 Jun 12. Mol Cell Proteomics. 2014. PMID: 24925906 Free PMC article.
References
-
- McCannel CA, Holland GN, Helm CJ, Cornell PJ, Winston JV, Rimmer TG. Causes of uveitis in the general practice of ophthalmology: UCLA Community-Based Uveitis Study Group. Am J Ophthalmol. 1996;121:35–46 - PubMed
-
- Hooper C, McCluskey P. Intraocular inflammation: its causes and investigations. Curr Allergy Asthma Rep. 2008;8:331–338 - PubMed
-
- Holland GN. Reconsidering the pathogenesis of ocular toxoplasmosis. Am J Ophthalmol. 1999;128:502–505 - PubMed
-
- Jones JL, Dargelas V, Roberts J, Press C, Remington JS, Montoya JG. Risk factors for Toxoplasma gondii infection in the United States. Clin Infect Dis. 2009;49:878–884 - PubMed
-
- Dubey JP, Jones JL. Toxoplasma gondii infection in humans and animals in the United States. Int J Parasitol. 2008;38:1257–1278 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

