The potential for caspases in drug discovery
- PMID: 20812148
- PMCID: PMC3289102
The potential for caspases in drug discovery
Abstract
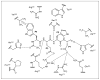
Caspases are a family of proteases that are involved in the execution of apoptosis and the inflammatory response. A plethora of diseases occur as a result of the dysregulation of apoptosis and inflammation, and caspases have been targeted as a therapeutic strategy to halt the progression of such diseases. Hundreds of peptide and peptidomimetic inhibitors have been designed and tested, but only a few have advanced to clinical trials because of poor drug-like properties and pharmacological constraints. Although much effort has been focused on inhibiting caspases, there are many diseases that result from a decrease in apoptosis, thus activating procaspases could also be a viable therapeutic strategy. To this end, recent efforts have focused on the design of procaspase-3 activators. This review highlights the current progress in the rational design of both specific and pan-caspase inhibitors, as well as procaspase-3 activators.
Figures


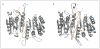
Similar articles
-
Small Molecule Active Site Directed Tools for Studying Human Caspases.Chem Rev. 2015 Nov 25;115(22):12546-629. doi: 10.1021/acs.chemrev.5b00434. Epub 2015 Nov 9. Chem Rev. 2015. PMID: 26551511 Free PMC article. Review.
-
Allosteric modulation of caspases.Pharmacol Ther. 2011 Nov;132(2):180-95. doi: 10.1016/j.pharmthera.2011.07.003. Epub 2011 Jul 23. Pharmacol Ther. 2011. PMID: 21807025 Review.
-
Small-molecule procaspase activators identified using fluorescence polarization.Chembiochem. 2013 Aug 19;14(12):1419-22. doi: 10.1002/cbic.201300315. Epub 2013 Jul 8. Chembiochem. 2013. PMID: 23836614
-
Turning on caspases with genetics and small molecules.Methods Enzymol. 2014;544:179-213. doi: 10.1016/B978-0-12-417158-9.00008-X. Methods Enzymol. 2014. PMID: 24974291 Free PMC article. Review.
-
A bifunctional allosteric site in the dimer interface of procaspase-3.Biophys Chem. 2011 Nov;159(1):100-9. doi: 10.1016/j.bpc.2011.05.013. Epub 2011 May 25. Biophys Chem. 2011. PMID: 21645959 Free PMC article.
Cited by
-
Selenium compounds, apoptosis and other types of cell death: an overview for cancer therapy.Int J Mol Sci. 2012;13(8):9649-9672. doi: 10.3390/ijms13089649. Epub 2012 Aug 2. Int J Mol Sci. 2012. PMID: 22949823 Free PMC article. Review.
-
Prevention of apoptosis averts glomerular tubular disconnection and podocyte loss in proteinuric kidney disease.Kidney Int. 2016 Jul;90(1):135-48. doi: 10.1016/j.kint.2016.03.026. Epub 2016 May 21. Kidney Int. 2016. PMID: 27217195 Free PMC article.
-
Role of Pyroptosis in Respiratory Diseases and its Therapeutic Potential.J Inflamm Res. 2022 Mar 28;15:2033-2050. doi: 10.2147/JIR.S352563. eCollection 2022. J Inflamm Res. 2022. PMID: 35370413 Free PMC article. Review.
-
Dehydroisohispanolone as a Promising NLRP3 Inhibitor Agent: Bioevaluation and Molecular Docking.Pharmaceuticals (Basel). 2022 Jul 2;15(7):825. doi: 10.3390/ph15070825. Pharmaceuticals (Basel). 2022. PMID: 35890124 Free PMC article.
-
De Novo Molecular Design of Caspase-6 Inhibitors by a GRU-Based Recurrent Neural Network Combined with a Transfer Learning Approach.Pharmaceuticals (Basel). 2021 Nov 30;14(12):1249. doi: 10.3390/ph14121249. Pharmaceuticals (Basel). 2021. PMID: 34959651 Free PMC article.
References
-
- Lamkanfi M, Festjens N, Declercq W, Vanden Berghe T, Vandenabeele P. Caspases in cell survival, proliferation and differentiation. Cell Death Differ. 2007;14(1):44–55. - PubMed
-
- Festjens N, Cornelis S, Lamkanfi M, Vandenabeele P. Caspase-containing complexes in the regulation of cell death and inflammation. Biol Chem. 2006;387(8):1005–1016. - PubMed
-
- Fleischer A, Ghadiri A, Dessauge F, Duhamer M, Rebollo MP, Alverez-Franco F, Rebollo A. Modulating apoptosis as a target for effective therapy. Mol Immunol. 2006;43(8):1065–1079. - PubMed
-
- Narula J, Chandrasekhar Y, Dec G. Apoptosis in heart failure: A tale of heightened expectations, unfulfilled promises and broken hearts…. Apoptosis. 1998;3(5):309–315. - PubMed
-
- Saraste A, Pulkki K, Kallajoki M, Henriksen K, Parvinen M, Voipio-Pulkki LM. Apoptosis in human acute myocardial infarction. Circulation. 1997;95(2):320–323. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
