Experience-dependent development of vocalization selectivity in the auditory cortex
- PMID: 20815478
- PMCID: PMC2945755
- DOI: 10.1121/1.3377057
Experience-dependent development of vocalization selectivity in the auditory cortex
Abstract
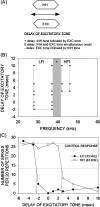
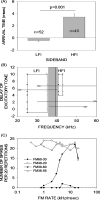
Vocalization-selective neurons are present in the auditory systems of several vertebrate groups. Vocalization selectivity is influenced by developmental experience, but the underlying mechanisms are only beginning to be understood. Evidence is presented in this review for the hypothesis that plasticity of timing and strength of inhibition is a mechanism for plasticity of vocalization selectivity. The pallid bat echolocates using downward frequency modulated (FM) sweeps. Nearly 70% of neurons with tuning in the echolocation frequency range in its auditory cortex respond selectively to the direction and rate of change of frequencies present in the echolocation call. During development, FM rate selectivity matures early, while direction selectivity emerges later. Based on the time course of development it was hypothesized that FM direction, but not rate, selectivity is experience-dependent. This hypothesis was tested by altering echolocation experience during development. The results show that normal echolocation experience is required for both refinement and maintenance of direction selectivity. Interestingly, experience is required for the maintenance of rate selectivity, but not for initial development. Across all ages and experimental groups, the timing relationship between inhibitory and excitatory inputs explains sweep selectivity. These experiments suggest that inhibitory plasticity is a substrate for experience-dependent changes in vocalization selectivity.
Figures

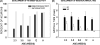

Similar articles
-
Neural mechanisms underlying selectivity for the rate and direction of frequency-modulated sweeps in the auditory cortex of the pallid bat.J Neurophysiol. 2006 Sep;96(3):1303-19. doi: 10.1152/jn.00020.2006. Epub 2006 Jun 14. J Neurophysiol. 2006. PMID: 16775213
-
Topography of sound level representation in the FM sweep selective region of the pallid bat auditory cortex.Hear Res. 2018 Sep;367:137-148. doi: 10.1016/j.heares.2018.05.017. Epub 2018 May 26. Hear Res. 2018. PMID: 29853324
-
Matched Behavioral and Neural Adaptations for Low Sound Level Echolocation in a Gleaning Bat, Antrozous pallidus.eNeuro. 2017 Mar 2;4(1):ENEURO.0018-17.2017. doi: 10.1523/ENEURO.0018-17.2017. eCollection 2017 Jan-Feb. eNeuro. 2017. PMID: 28275715 Free PMC article.
-
Multiple mechanisms shape selectivity for FM sweep rate and direction in the pallid bat inferior colliculus and auditory cortex.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2011 May;197(5):615-23. doi: 10.1007/s00359-010-0554-0. Epub 2010 Jul 2. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2011. PMID: 20596868 Free PMC article. Review.
-
Neuroethology of auditory cortex.Jpn J Physiol. 1991;41(5):671-91. doi: 10.2170/jjphysiol.41.671. Jpn J Physiol. 1991. PMID: 1803053 Review. No abstract available.
Cited by
-
Phonological processing in human auditory cortical fields.Front Hum Neurosci. 2011 Apr 20;5:42. doi: 10.3389/fnhum.2011.00042. eCollection 2011. Front Hum Neurosci. 2011. PMID: 21541252 Free PMC article.
-
FM-selective networks in human auditory cortex revealed using fMRI and multivariate pattern classification.J Cogn Neurosci. 2012 Sep;24(9):1896-907. doi: 10.1162/jocn_a_00254. Epub 2012 May 29. J Cogn Neurosci. 2012. PMID: 22640390 Free PMC article.
-
Understanding the neurophysiological basis of auditory abilities for social communication: a perspective on the value of ethological paradigms.Hear Res. 2013 Nov;305:3-9. doi: 10.1016/j.heares.2013.08.008. Epub 2013 Aug 27. Hear Res. 2013. PMID: 23994815 Free PMC article.
References
-
- Bell, G. P. (1982). “Behavioral and ecological aspects of gleaning by the desert insectivorous bat, Antrozous pallidus (Chiroptera: Vespertilionidae),” Behav. Ecol. Sociobiol. BESOD6 10, 217–223.10.1007/BF00299688 - DOI
-
- Brosch, M., and Schreiner, C. E. (1997). “Time course of forward masking tuning curves in cat primary auditory cortex,” J. Neurophysiol. JONEA4 77, 923–943. - PubMed
-
- Brown, P. (1976). “Vocal communication in the pallid bat, Antrozous pallidus,” Z. Tierpsychol ZETIAG 41, 34–54. - PubMed
-
- Brown, P., Grinnell, A. D., and Harrison, J. (1978). “The development of hearing in the pallid bat, Antrozous pallidus,” J. Comp. Physiol. [A] 126, 169–182.10.1007/BF00666371 - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

