Altered expression of regulators of the cortical chloride transporters NKCC1 and KCC2 in schizophrenia
- PMID: 20819979
- PMCID: PMC3015012
- DOI: 10.1001/archgenpsychiatry.2010.114
Altered expression of regulators of the cortical chloride transporters NKCC1 and KCC2 in schizophrenia
Abstract
Context: Disturbances in markers of cortical γ-aminobutyric acid neurotransmission are a common finding in schizophrenia. The nature of γ-aminobutyric acid neurotransmission (hyperpolarizing or depolarizing) depends on the local intracellular chloride concentration. In the central nervous system, the intracellular chloride level is determined by the activity of 2 cation-chloride transporters, NKCC1 and KCC2. The activities of these transporters are in turn regulated by a network of serine-threonine kinases that includes OXSR1, STK39, and the WNK kinases WNK1, WNK3, and WNK4.
Objective: To compare the levels of NKCC1, KCC2, OXSR1, STK39, WNK1, WNK3, and WNK4 transcripts in prefrontal cortex area 9 between subjects with schizophrenia and healthy comparison subjects.
Design: Real-time quantitative polymerase chain reaction technique was used to measure transcript levels in the prefrontal cortex.
Setting: Human brain specimens were obtained from autopsies conducted at the Allegheny County Medical Examiner's Office, Pittsburgh, Pennsylvania.
Participants: Postmortem brain specimens from 42 subjects with schizophrenia and 42 matched healthy comparison subjects. Brain specimens from 18 macaque monkeys exposed to haloperidol, olanzapine, or sham long-term.
Main outcome measures: Relative expression levels for NKCC1, KCC2, OXSR1, STK39, WNK1, WNK3, and WNK4 transcripts compared with the mean expression level of 3 housekeeping transcripts.
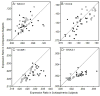
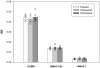
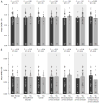
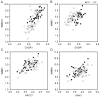
Results: OXSR1 and WNK3 transcripts were substantially overexpressed in subjects with schizophrenia relative to comparison subjects. In contrast, NKCC1, KCC2, STK39, WNK1, and WNK4 transcript levels did not differ between subject groups. OXSR1 and WNK3 transcript expression levels were not changed in antipsychotic-exposed monkeys and were not affected by potential confounding factors in the subjects with schizophrenia.
Conclusion: In schizophrenia, increased expression levels, and possibly increased kinase activities, of OXSR1 and WNK3 may shift the balance of chloride transport by NKCC1 and KCC2 and alter the nature of γ-aminobutyric acid neurotransmission in the prefrontal cortex.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

