Intracellular trafficking of silicon particles and logic-embedded vectors
- PMID: 20820744
- PMCID: PMC2936484
- DOI: 10.1039/c0nr00227e
Intracellular trafficking of silicon particles and logic-embedded vectors
Abstract
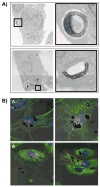
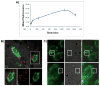
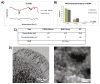
Mesoporous silicon particles show great promise for use in drug delivery and imaging applications as carriers for second-stage nanoparticles and higher order particles or therapeutics. Modulation of particle geometry, surface chemistry, and porosity allows silicon particles to be optimized for specific applications such as vascular targeting and avoidance of biological barriers commonly found between the site of drug injection and the final destination. In this study, the intracellular trafficking of unloaded carrier silicon particles and carrier particles loaded with secondary iron oxide nanoparticles was investigated. Following cellular uptake, membrane-encapsulated silicon particles migrated to the perinuclear region of the cell by a microtubule-driven mechanism. Surface charge, shape (spherical and hemispherical) and size (1.6 and 3.2 microm) of the particle did not alter the rate of migration. Maturation of the phagosome was associated with an increase in acidity and acquisition of markers of late endosomes and lysosomes. Cellular uptake of iron oxide nanoparticle-loaded silicon particles resulted in sorting of the particles and trafficking to unique destinations. The silicon carriers remained localized in phagosomes, while the second stage iron oxide nanoparticles were sorted into multi-vesicular bodies that dissociated from the phagosome into novel membrane-bound compartments. Release of iron from the cells may represent exocytosis of iron oxide nanoparticle-loaded vesicles. These results reinforce the concept of multi-functional nanocarriers, in which different particles are able to perform specific tasks, in order to deliver single- or multi-component payloads to specific sub-cellular compartments.
Figures



Similar articles
-
Logic-embedded vectors for intracellular partitioning, endosomal escape, and exocytosis of nanoparticles.Small. 2010 Dec 6;6(23):2691-700. doi: 10.1002/smll.201000727. Epub 2010 Oct 18. Small. 2010. PMID: 20957619 Free PMC article.
-
Mitotic trafficking of silicon microparticles.Nanoscale. 2009 Nov;1(2):250-9. doi: 10.1039/b9nr00138g. Epub 2009 Oct 5. Nanoscale. 2009. PMID: 20644846 Free PMC article.
-
Multi-stage delivery nano-particle systems for therapeutic applications.Biochim Biophys Acta. 2011 Mar;1810(3):317-29. doi: 10.1016/j.bbagen.2010.05.004. Epub 2010 May 21. Biochim Biophys Acta. 2011. PMID: 20493927 Free PMC article. Review.
-
Near-infrared light and magnetic field dual-responsive porous silicon-based nanocarriers to overcome multidrug resistance in breast cancer cells with enhanced efficiency.J Mater Chem B. 2020 Jan 22;8(3):546-557. doi: 10.1039/c9tb02340b. J Mater Chem B. 2020. PMID: 31854435
-
Role of microtubules and myosins in Fc gamma receptor-mediated phagocytosis.Front Biosci. 2006 May 1;11:1479-90. doi: 10.2741/1897. Front Biosci. 2006. PMID: 16368530 Review.
Cited by
-
Cellular communication via nanoparticle-transporting biovesicles.Nanomedicine (Lond). 2014 Apr;9(5):581-592. doi: 10.2217/nnm.13.57. Epub 2013 Jun 3. Nanomedicine (Lond). 2014. PMID: 23731456 Free PMC article.
-
The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles.Int J Nanomedicine. 2012;7:5577-91. doi: 10.2147/IJN.S36111. Epub 2012 Nov 2. Int J Nanomedicine. 2012. PMID: 23144561 Free PMC article. Review.
-
Evaluation of cell function upon nanovector internalization.Small. 2013 May 27;9(9-10):1696-702. doi: 10.1002/smll.201202001. Epub 2012 Nov 20. Small. 2013. PMID: 23166049 Free PMC article.
-
A pyruvate decarboxylase-mediated therapeutic strategy for mimicking yeast metabolism in cancer cells.Pharmacol Res. 2016 Sep;111:413-421. doi: 10.1016/j.phrs.2016.07.005. Epub 2016 Jul 6. Pharmacol Res. 2016. PMID: 27394167 Free PMC article.
-
Endolysosomal Mesoporous Silica Nanoparticle Trafficking along Microtubular Highways.Pharmaceutics. 2021 Dec 27;14(1):56. doi: 10.3390/pharmaceutics14010056. Pharmaceutics. 2021. PMID: 35056951 Free PMC article.
References
-
- Ferrari M. Nat Rev Cancer. 2005;5:161–171. - PubMed
-
- Torchilin VP. Adv Drug Deliv Rev. 2006;58:1532–1555. - PubMed
-
- Tasciotti E, Liu X, Bhavane R, Plant K, Leonard AD, Price BK, Cheng MMC, Decuzzi P, Tour JM, Robertson F, Ferrari M. Nat Nano. 2008;3:151–157. - PubMed
-
- Decuzzi P, Ferrari M. Biomaterials. 2008;29:377–384. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

