Early and sustained increase in the expression of hippocampal IGF-1, but not EPO, in a developmental rodent model of traumatic brain injury
- PMID: 20822461
- PMCID: PMC6468946
- DOI: 10.1089/neu.2009.1226
Early and sustained increase in the expression of hippocampal IGF-1, but not EPO, in a developmental rodent model of traumatic brain injury
Abstract
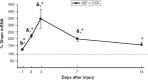
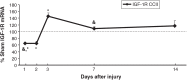
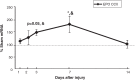
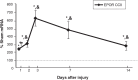
Pediatric traumatic brain injury (pTBI) is the leading cause of traumatic death and disability in children in the United States. Impaired learning and memory in these young survivors imposes a heavy toll on society. In adult TBI (aTBI) models, cognitive outcome improved after administration of erythropoietin (EPO) or insulin-like growth factor-1 (IGF-1). Little is known about the production of these agents in the hippocampus, a brain region critical for learning and memory, after pTBI. Our objective was to describe hippocampal expression of EPO and IGF-1, together with their receptors (EPOR and IGF-1R, respectively), over time after pTBI in 17-day-old rats. We used the controlled cortical impact (CCI) model and measured hippocampal mRNA levels of EPO, IGF-1, EPOR, IGF-1R, and markers of caspase-dependent apoptosis (bcl2, bax, and p53) at post-injury days (PID) 1, 2, 3, 7, and 14. CCI rats performed poorly on Morris water maze testing of spatial working memory, a hippocampally-based cognitive function. Apoptotic markers were present early and persisted for the duration of the study. EPO in our pTBI model increased much later (PID7) than in aTBI models (12 h), while EPOR and IGF-1 increased at PID1 and PID2, respectively, similar to data from aTBI models. Our data indicate that EPO expression showed a delayed upregulation post-pTBI, while EPOR increased early. We speculate that administration of EPO in the first 1-2 days after pTBI would increase hippocampal neuronal survival and function.
Figures




References
-
- Adelson P.D. Whalen M.J. Kochanek P.M. Robichaud P. Carlos T.M. Blood brain barrier permeability and acute inflammation in two models of traumatic brain injury in the immature rat: a preliminary report. Acta Neurochir. Suppl. 1998;71:104–106. - PubMed
-
- Agarwal M.K. Agarwal M.L. Athar M. Gupta S. Tocotrienol-rich fraction of palm oil activates p53, modulates Bax/Bcl2 ratio and induces apoptosis independent of cell cycle association. Cell Cycle (Georgetown, Tex 3) 2004. pp. 205–211. - PubMed
-
- Anderson V. Catroppa C. Morse S. Haritou F. Rosenfeld J. Functional plasticity or vulnerability after early brain injury? Pediatrics. 2005;116:1374–1382. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

