Interplay of mechanical and binding properties of Fibronectin type I
- PMID: 20824113
- PMCID: PMC2932639
- DOI: 10.1007/s00214-009-0677-y
Interplay of mechanical and binding properties of Fibronectin type I
Abstract
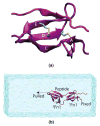
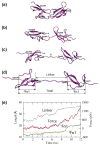
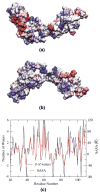
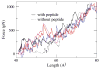
Fibronectins (FNs) are a major component of the extracellular matrix (ECM), and provide important binding sites for a variety of ligands outside and on the surface of the cell. Similar to other ECM proteins, FNs are consistently subject to mechanical stress in the ECM. Therefore, it is important to study their structure and binding properties under mechanical stress and understand how their binding and mechanical properties might affect each other. Although certain FN modules have been extensively investigated, no simulation studies have been reported for the FN type I (Fn1) domains, despite their prominent role in binding of various protein modules to FN polymers in the ECM. Using equilibrium and steered molecular dynamics simulations, we have studied mechanical properties of Fn1 modules in the presence or the absence of a specific FN-binding peptide (FnBP). We have also investigated how the binding of the FnBP peptide to Fn1 might be affected by tensile force. Despite the presence of disulfide bonds within individual Fn1 modules that are presumed to prevent their extension, it is found that significant internal structural changes within individual modules are induced by the forces applied in our simulations. These internal structural changes result in significant variations in the accessibility of different residues of the Fn1 modules, which affect their exposure, and, thus, the binding properties of the Fn1 modules. Binding of the FnBP appears to reduce the flexibility of the linker region connecting individual Fn1 modules (exhibited in the form of reduced fluctuation and motion of the linker region), both with regard to bending and stretching motions, and hence stabilizes the inter-domain configuration under force. Under large tensile forces, the FnBP peptide unbinds from Fn1. The results suggest that Fn1 modules in FN polymers do contribute to the overall extension caused by force-induced stretching of the polymer in the ECM, and that binding properties of Fn1 modules can be affected by mechanically induced internal protein conformational changes in spite of the presence of disulfide bonds which were presumed to completely abolish the capacity of Fn1 modules to undergo extension in response to external forces.
Figures


Similar articles
-
Fibronectin forms the most extensible biological fibers displaying switchable force-exposed cryptic binding sites.Proc Natl Acad Sci U S A. 2009 Oct 27;106(43):18267-72. doi: 10.1073/pnas.0907518106. Epub 2009 Oct 13. Proc Natl Acad Sci U S A. 2009. PMID: 19826086 Free PMC article.
-
Transglutaminase 2 has higher affinity for relaxed than for stretched fibronectin fibers.Matrix Biol. 2024 Jan;125:113-132. doi: 10.1016/j.matbio.2023.12.006. Epub 2023 Dec 20. Matrix Biol. 2024. PMID: 38135164
-
Sequence and structural analysis of fibronectin-binding protein reveals importance of multiple intrinsic disordered tandem repeats.J Mol Recognit. 2019 Apr;32(4):e2768. doi: 10.1002/jmr.2768. Epub 2018 Nov 5. J Mol Recognit. 2019. PMID: 30397967
-
Fibronectin Mechanobiology Regulates Tumorigenesis.Cell Mol Bioeng. 2016;9:1-11. doi: 10.1007/s12195-015-0417-4. Epub 2015 Aug 15. Cell Mol Bioeng. 2016. PMID: 26900407 Free PMC article. Review.
-
Multiscale relationships between fibronectin structure and functional properties.Acta Biomater. 2014 Apr;10(4):1524-31. doi: 10.1016/j.actbio.2013.08.027. Epub 2013 Aug 24. Acta Biomater. 2014. PMID: 23978411 Review.
Cited by
-
The cardiac nanoenvironment: form and function at the nanoscale.Biophys Rev. 2021 Aug 31;13(5):625-636. doi: 10.1007/s12551-021-00834-5. eCollection 2021 Oct. Biophys Rev. 2021. PMID: 34765045 Free PMC article. Review.
-
Microarray analysis of perinatal-estrogen-induced changes in gene expression related to brain sexual differentiation in mice.PLoS One. 2013 Nov 4;8(11):e79437. doi: 10.1371/journal.pone.0079437. eCollection 2013. PLoS One. 2013. PMID: 24223949 Free PMC article.
-
Extracellular Matrix Structure and Composition in the Early Four-Chambered Embryonic Heart.Cells. 2020 Jan 24;9(2):285. doi: 10.3390/cells9020285. Cells. 2020. PMID: 31991580 Free PMC article.
-
Revealing Atomic-Level Mechanisms of Protein Allostery with Molecular Dynamics Simulations.PLoS Comput Biol. 2016 Jun 10;12(6):e1004746. doi: 10.1371/journal.pcbi.1004746. eCollection 2016 Jun. PLoS Comput Biol. 2016. PMID: 27285999 Free PMC article. Review.
-
Stretching fibronectin fibres disrupts binding of bacterial adhesins by physically destroying an epitope.Nat Commun. 2010;1:135. doi: 10.1038/ncomms1135. Nat Commun. 2010. PMID: 21139580 Free PMC article.
References
-
- Ingber D. The architecure of life. Scientific American. 1998 January;:48–57. - PubMed
-
- Humphries MJ, Obara M, Yamada KOKM. Role of Fibronectin in addhesion, migration, and metastasis. Cancer Invest. 1989;7:373–393. - PubMed
-
- Mosher DF. Fibronectin. Academic Press; New York: 1989.
-
- Hynes RO. Fibronectins. Springer; New York: 1990.
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
