γ-Glutamyl transpeptidase is a heavily N-glycosylated heterodimer in HepG2 cells
- PMID: 20831856
- PMCID: PMC2965302
- DOI: 10.1016/j.abb.2010.08.019
γ-Glutamyl transpeptidase is a heavily N-glycosylated heterodimer in HepG2 cells
Abstract
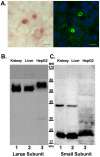
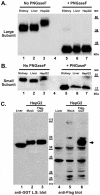
The cell surface enzyme γ-glutamyl transpeptidase (GGT) is expressed by human hepatocellular carcinomas (HCCs). HCCs arise from malignant transformation of hepatocytes and are the most common form of primary liver cancer. Identification of tumor-specific, post-translational modifications of GGT may provide novel biomarkers for HCC. The HepG2 cell line, derived from a human HCC, has been used extensively in studies of liver cancer. However, the use of this cell line for studies of GGT have been stymied by reports that HepG2 cells do not process the GGT propeptide into its heterodimeric subunits. The data in this study demonstrate that HepG2 cells do, in fact, produce the mature heterodimeric form of GGT. Immunohistochemical and immunoaffinity analyses provide direct evidence that, in HepG2 cells, GGT is properly localized to the bile canaliculi. Three independent, experimental approaches demonstrate that GGT in HepG2 cells is comprised of two subunits that are more heavily N-glycosylated than GGT from normal human liver tissue. These data directly contradict the dogma in the field. These data support the use of HepG2 cells as a model system for analyzing tumor-specific changes in the post-translational modifications of GGT.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Autocatalytic cleavage of human gamma-glutamyl transpeptidase is highly dependent on N-glycosylation at asparagine 95.J Biol Chem. 2011 Aug 19;286(33):28876-28888. doi: 10.1074/jbc.M111.248823. Epub 2011 Jun 28. J Biol Chem. 2011. PMID: 21712391 Free PMC article.
-
gamma-Glutamyl transpeptidase-dependent lipid peroxidation in isolated hepatocytes and HepG2 hepatoma cells.Free Radic Biol Med. 1997;22(5):853-60. doi: 10.1016/s0891-5849(96)00422-4. Free Radic Biol Med. 1997. PMID: 9119254
-
Gamma-glutamyltransferase from human hepatoma cell lines: purification and cell culture of HepG2 on microcarriers.Clin Chim Acta. 1990 Nov 5;191(3):221-32. doi: 10.1016/0009-8981(90)90023-l. Clin Chim Acta. 1990. PMID: 1979762
-
Structural, functional, and clinical aspects of gamma-glutamyltransferase.CRC Crit Rev Clin Lab Sci. 1980;12(1):1-58. doi: 10.3109/10408368009108725. CRC Crit Rev Clin Lab Sci. 1980. PMID: 6104563 Review.
-
Targeting gamma-glutamyl transpeptidase: A pleiotropic enzyme involved in glutathione metabolism and in the control of redox homeostasis.Free Radic Biol Med. 2023 Nov 1;208:672-683. doi: 10.1016/j.freeradbiomed.2023.09.020. Epub 2023 Sep 20. Free Radic Biol Med. 2023. PMID: 37739139 Review.
Cited by
-
Glutaminolysis is Essential for Energy Production and Ion Transport in Human Corneal Endothelium.EBioMedicine. 2017 Feb;16:292-301. doi: 10.1016/j.ebiom.2017.01.004. Epub 2017 Jan 13. EBioMedicine. 2017. PMID: 28117276 Free PMC article.
-
A γ-Glutamyl Transpeptidase (GGT)-Triggered Charge Reversal Drug-Delivery System for Cervical Cancer Treatment: In Vitro and In Vivo Investigation.Pharmaceutics. 2023 Apr 25;15(5):1335. doi: 10.3390/pharmaceutics15051335. Pharmaceutics. 2023. PMID: 37242579 Free PMC article.
-
Marine-Inspired Ovothiol Analogs Inhibit Membrane-Bound Gamma-Glutamyl-Transpeptidase and Modulate Reactive Oxygen Species and Glutathione Levels in Human Leukemic Cells.Mar Drugs. 2025 Jul 30;23(8):308. doi: 10.3390/md23080308. Mar Drugs. 2025. PMID: 40863625 Free PMC article.
-
Engineered extracellular matrices with controlled mechanics modulate renal proximal tubular cell epithelialization.PLoS One. 2017 Jul 17;12(7):e0181085. doi: 10.1371/journal.pone.0181085. eCollection 2017. PLoS One. 2017. PMID: 28715434 Free PMC article.
-
Application of glycoproteomics for the discovery of biomarkers in lung cancer.Proteomics Clin Appl. 2012 Jun;6(5-6):244-56. doi: 10.1002/prca.201100042. Proteomics Clin Appl. 2012. PMID: 22641610 Free PMC article. Review.
References
-
- Hanigan MH, Ricketts WA. Extracellular glutathione is a source of cysteine for cells that express gamma-glutamyl transpeptidase. Biochemistry. 1993;32:6302–6306. - PubMed
-
- Ikeda Y, Taniguchi N. Gene expression of gamma-glutamyltranspeptidase. Methods Enzymol. 2005;401:408–425. - PubMed
-
- Yao DF, Dong ZZ. Hepatoma-related gamma-glutamyl transferase in laboratory or clinical diagnosis of hepatocellular carcinoma. Hepatobiliary Pancreat Dis Int. 2007;6:9–11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

