An Smc3 acetylation cycle is essential for establishment of sister chromatid cohesion
- PMID: 20832721
- PMCID: PMC4766734
- DOI: 10.1016/j.molcel.2010.08.008
An Smc3 acetylation cycle is essential for establishment of sister chromatid cohesion
Abstract
Sister chromatid cohesion is thought to involve entrapment of sister DNAs by a tripartite ring composed of the cohesin subunits Smc1, Smc3, and Scc1. Establishment of cohesion during S phase depends on acetylation of Smc3's nucleotide-binding domain (NBD) by the Eco1 acetyl transferase. It is destroyed at the onset of anaphase due to Scc1 cleavage by separase. In yeast, Smc3 acetylation is reversed at anaphase by the Hos1 deacetylase as a consequence of Scc1 cleavage. Smc3 molecules that remain acetylated after mitosis due to Hos1 inactivation cannot generate cohesion during the subsequent S phase, implying that cohesion establishment depends on de novo acetylation during DNA replication. By inducing Smc3 deacetylation in postreplicative cells due to Hos1 overexpression, we provide evidence that Smc3 acetylation contributes to the maintenance of sister chromatid cohesion. A cycle of Smc3 NBD acetylation is therefore an essential aspect of the chromosome cycle in eukaryotic cells.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures
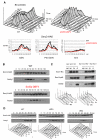



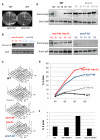
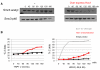

Comment in
-
Recycling cohesin rings by deacetylation.Mol Cell. 2010 Sep 10;39(5):657-9. doi: 10.1016/j.molcel.2010.08.032. Mol Cell. 2010. PMID: 20832715
-
Cell cycle: cycling through acetylation.Nat Rev Mol Cell Biol. 2010 Nov;11(11):755. doi: 10.1038/nrm3002. Nat Rev Mol Cell Biol. 2010. PMID: 20966967 No abstract available.
References
-
- Rolef Ben-Shahar T, Heeger S, Lehane C, East P, Flynn H, Skehel M, Uhlmann F. Eco1-dependent cohesin acetylation during establishment of sister chromatid cohesion. Science. 2008;321:563–566. - PubMed
-
- Ciosk R, Shirayama M, Shevchenko A, Tanaka T, Toth A, Shevchenko A, Nasmyth K. Cohesin’s binding to chromosomes depends on a separate complex consisting of Scc2 and Scc4 proteins. Mol. Cell. 2000;5:243–254. - PubMed
-
- Davie JK, Edmondson DG, Coco CB, Dent SY. Tup1-Ssn6 interacts with multiple class I histone deacetylases in vivo. J. Biol. Chem. 2003;278:50158–50162. - PubMed
-
- Gruber S, Haering CH, Nasmyth K. Chromosomal cohesin forms a ring. Cell. 2003;112:765–777. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

