Targeting mitochondrial glutaminase activity inhibits oncogenic transformation
- PMID: 20832749
- PMCID: PMC3078749
- DOI: 10.1016/j.ccr.2010.08.009
Targeting mitochondrial glutaminase activity inhibits oncogenic transformation
Erratum in
- Cancer Cell. 2010 Oct 19;18(4):397
Abstract
Rho GTPases impact a number of activities important for oncogenesis. We describe a small molecule inhibitor that blocks oncogenic transformation induced by various Rho GTPases in fibroblasts, and the growth of human breast cancer and B lymphoma cells, without affecting normal cells. We identify the target of this inhibitor to be the metabolic enzyme glutaminase, which catalyzes the hydrolysis of glutamine to glutamate. We show that transformed fibroblasts and breast cancer cells exhibit elevated glutaminase activity that is dependent on Rho GTPases and NF-κB activity, and is blocked by the small molecule inhibitor. These findings highlight a previously unappreciated connection between Rho GTPase activation and cellular metabolism and demonstrate that targeting glutaminase activity can inhibit oncogenic transformation.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures
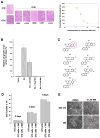
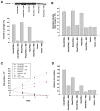


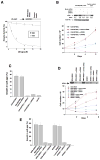

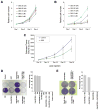

Comment on
-
Cancer metabolism: is glutamine sweeter than glucose?Cancer Cell. 2010 Sep 14;18(3):199-200. doi: 10.1016/j.ccr.2010.08.017. Cancer Cell. 2010. PMID: 20832746 Free PMC article.
References
-
- Baird D, Feng Q, Cerione RA. The Cool-2/α-Pix protein mediates a Cdc42-Rac signaling cascade. Current Biology. 2005;15:1–10. - PubMed
-
- Burbelo P, Wellstein A, Pestell RG. Altered Rho GTPase signaling pathways in breast cancer cells. Breast Cancer Res Treat. 2004;84:43–48. - PubMed
-
- Cammarano MS, Minden A. Dbl and the Rho GTPases activate NFκB by IκB kinase (IKK)-dependent and IKK-independent pathways. J Biol Chem. 2001;276:25876–25882. - PubMed
-
- Christofk HR, Vander Heiden MG, Wu N, Asara JM, Cantley LC. Pyruvate kinase M2 is a phosphotyrosine-binding protein. Nature. 2008a;452:181–186. - PubMed
-
- Christofk HR, Vander Heiden MG, Harris MH, Ramanathan A, Gerszten RE, Wei R, Fleming MD, Schreiber SL, Cantley LC. The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature. 2008b;452:230–233. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

