Virus-bacterium coupling driven by both turbidity and hydrodynamics in an Amazonian floodplain lake
- PMID: 20833790
- PMCID: PMC2976232
- DOI: 10.1128/AEM.01161-10
Virus-bacterium coupling driven by both turbidity and hydrodynamics in an Amazonian floodplain lake
Abstract
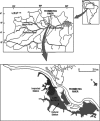
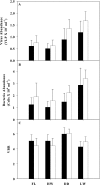
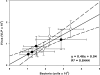
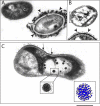
The importance of viruses in aquatic ecosystem functioning has been widely described. However, few studies have examined tropical aquatic ecosystems. Here, we evaluated for the first time viruses and their relationship with other planktonic communities in an Amazonian freshwater ecosystem. Coupling between viruses and bacteria was studied, focusing both on hydrologic dynamics and anthropogenic forced turbidity in the system (Lake Batata). Samples were taken during four hydrologic seasons at both natural and impacted sites to count virus-like particles (VLP) and bacteria. In parallel, virus-infected bacteria were identified and quantified by transmission electron microscopy (TEM). Viral abundance ranged from 0.5 × 10⁷ ± 0.2 × 10⁷ VLP ml⁻¹ (high-water season, impacted site) to 1.7 × 10⁷ ± 0.4 × 10⁷ VLP ml⁻¹ (low-water season, natural site). These data were strongly correlated with the bacterial abundance (r² = 0.84; P < 0.05), which ranged from 1.0 × 10⁶ ± 0.5 × 10⁶ cells ml⁻¹ (high water, impacted site) to 3.4 × 10⁶ ± 0.7 × 10⁶ cells ml⁻¹ (low water, natural site). Moreover, the viral abundance was weakly correlated with chlorophyll a, suggesting that most viruses were bacteriophages. TEM quantitative analyses revealed that the frequency of visibly infected cells was 20%, with 10 ± 3 phages per cell section. In general, we found a low virus-bacterium ratio (<7). Both the close coupling between the viral and bacterial abundances and the low virus-bacterium ratio suggest that viral abundance tends to be driven by the reduction of hosts for viral infection. Our results demonstrate that viruses are controlled by biological substrates, whereas in addition to grazing, bacteria are regulated by physical processes caused by turbidity, which affect underwater light distribution and dissolved organic carbon availability.
Figures
Similar articles
-
Bacteria and viruses in the water column of tropical freshwater reservoirs.Environ Microbiol. 2004 Jul;6(7):707-15. doi: 10.1111/j.1462-2920.2004.00602.x. Environ Microbiol. 2004. PMID: 15186349
-
Viral abundance, production, decay rates and life strategies (lysogeny versus lysis) in Lake Bourget (France).Environ Microbiol. 2011 Mar;13(3):616-30. doi: 10.1111/j.1462-2920.2010.02364.x. Epub 2010 Nov 5. Environ Microbiol. 2011. PMID: 21054737
-
Virus-bacterium interactions in water and sediment of West African inland aquatic systems.Appl Environ Microbiol. 2006 Aug;72(8):5274-82. doi: 10.1128/AEM.00863-06. Appl Environ Microbiol. 2006. PMID: 16885276 Free PMC article.
-
Virioplankton: viruses in aquatic ecosystems.Microbiol Mol Biol Rev. 2000 Mar;64(1):69-114. doi: 10.1128/MMBR.64.1.69-114.2000. Microbiol Mol Biol Rev. 2000. PMID: 10704475 Free PMC article. Review.
-
[Virus and prophages in aquatic ecosystems].Can J Microbiol. 2009 Feb;55(2):95-109. doi: 10.1139/w08-099. Can J Microbiol. 2009. PMID: 19295641 Review. French.
Cited by
-
Virus ecology of fluvial systems: a blank spot on the map?Biol Rev Camb Philos Soc. 2016 Nov;91(4):937-949. doi: 10.1111/brv.12202. Epub 2015 Jun 24. Biol Rev Camb Philos Soc. 2016. PMID: 26105126 Free PMC article. Review.
-
Abundance and Distribution of Microbial Cells and Viruses in an Alluvial Aquifer.Front Microbiol. 2017 Jul 11;8:1199. doi: 10.3389/fmicb.2017.01199. eCollection 2017. Front Microbiol. 2017. PMID: 28744257 Free PMC article.
-
Virioplankton Assemblage Structure in the Lower River and Ocean Continuum of the Amazon.mSphere. 2017 Oct 4;2(5):e00366-17. doi: 10.1128/mSphere.00366-17. eCollection 2017 Sep-Oct. mSphere. 2017. PMID: 28989970 Free PMC article.
-
Viruses and bacteria in floodplain lakes along a major Amazon tributary respond to distance to the Amazon River.Front Microbiol. 2015 Mar 4;6:158. doi: 10.3389/fmicb.2015.00158. eCollection 2015. Front Microbiol. 2015. PMID: 25788895 Free PMC article.
-
Potential effects of UV radiation on photosynthetic structures of the bloom-forming cyanobacterium Cylindrospermopsis raciborskii CYRF-01.Front Microbiol. 2015 Oct 30;6:1202. doi: 10.3389/fmicb.2015.01202. eCollection 2015. Front Microbiol. 2015. PMID: 26579108 Free PMC article.
References
-
- Amado, A. M., J. B. Cotner, A. L. Suhett, F. Assis Esteves, R. L. Bozelli, and V. F. Farjalla. 2007. Contrasting interactions mediate dissolved organic matter decomposition in tropical aquatic ecosystems. Aquat. Microb. Ecol. 49:25-34.
-
- Amado, A. M., F. F. Vinicius, A. E. Francisco de, L. B. Reinaldo, R. Fábio, and E.-P. Alex. 2006. Complementary pathways of dissolved organic carbon removal pathways in clear-water Amazonian ecosystems: photochemical degradation and bacterial uptake. FEMS Microbiol. Ecol. 56:8-17. - PubMed
-
- Amon, R. M. W., and R. Benner. 1996. Bacterial utilization of different size classes of dissolved organic matter. Limnol. Oceanogr. 41:41-51.
-
- Anesio, A. M., P. C. Abreu, and F. D. Esteves. 1997. Influence of the hydrological cycle on the bacterioplankton of an impacted clear water Amazonian lake. Microb. Ecol. 34:66-73. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

