Differences in signalling by directly and indirectly binding ligands in bacterial chemotaxis
- PMID: 20834231
- PMCID: PMC2964171
- DOI: 10.1038/emboj.2010.224
Differences in signalling by directly and indirectly binding ligands in bacterial chemotaxis
Abstract
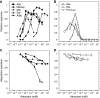
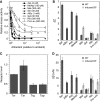
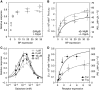
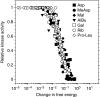
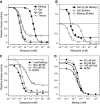
In chemotaxis of Escherichia coli and other bacteria, extracellular stimuli are perceived by transmembrane receptors that bind their ligands either directly, or indirectly through periplasmic-binding proteins (BPs). As BPs are also involved in ligand uptake, they provide a link between chemotaxis and nutrient utilization by cells. However, signalling by indirectly binding ligands remains much less understood than signalling by directly binding ligands. Here, we compared intracellular responses mediated by both types of ligands and developed a new mathematical model for signalling by indirectly binding ligands. We show that indirect binding allows cells to better control sensitivity to specific ligands in response to their nutrient environment and to coordinate chemotaxis with ligand transport, but at the cost of the dynamic range being much narrower than for directly binding ligands. We further demonstrate that signal integration by the chemosensory complexes does not depend on the type of ligand. Overall, our data suggest that the distinction between signalling by directly and indirectly binding ligands is more physiologically important than the traditional distinction between high- and low-abundance receptors.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures
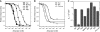
Comment in
-
Classifying chemoreceptors: quantity versus quality.EMBO J. 2010 Oct 20;29(20):3435-6. doi: 10.1038/emboj.2010.246. EMBO J. 2010. PMID: 20959858 Free PMC article.
Similar articles
-
Simultaneous high gain and wide dynamic range in a model of bacterial chemotaxis.IET Syst Biol. 2007 Jul;1(4):222-9. doi: 10.1049/iet-syb:20070003. IET Syst Biol. 2007. PMID: 17708429
-
Lateral density of receptor arrays in the membrane plane influences sensitivity of the E. coli chemotaxis response.EMBO J. 2011 May 4;30(9):1719-29. doi: 10.1038/emboj.2011.77. Epub 2011 Mar 25. EMBO J. 2011. PMID: 21441899 Free PMC article.
-
Chemical probes of bacterial signal transduction reveal that repellents stabilize and attractants destabilize the chemoreceptor array.ACS Chem Biol. 2008 Feb 15;3(2):101-9. doi: 10.1021/cb700211s. ACS Chem Biol. 2008. PMID: 18278851
-
Receptor clustering and signal processing in E. coli chemotaxis.Trends Microbiol. 2004 Dec;12(12):569-76. doi: 10.1016/j.tim.2004.10.003. Trends Microbiol. 2004. PMID: 15539117 Review.
-
pH sensing in bacterial chemotaxis.Novartis Found Symp. 1999;221:38-50; discussions 50-4. doi: 10.1002/9780470515631.ch4. Novartis Found Symp. 1999. PMID: 10207912 Review.
Cited by
-
Chemotaxis to the quorum-sensing signal AI-2 requires the Tsr chemoreceptor and the periplasmic LsrB AI-2-binding protein.J Bacteriol. 2011 Feb;193(3):768-73. doi: 10.1128/JB.01196-10. Epub 2010 Nov 19. J Bacteriol. 2011. PMID: 21097621 Free PMC article.
-
Activity, Abundance, and Localization of Quorum Sensing Receptors in Vibrio harveyi.Front Microbiol. 2017 Apr 18;8:634. doi: 10.3389/fmicb.2017.00634. eCollection 2017. Front Microbiol. 2017. PMID: 28458660 Free PMC article.
-
Spatiotemporal analysis of different mechanisms for interpreting morphogen gradients.Biophys J. 2015 Apr 21;108(8):2061-73. doi: 10.1016/j.bpj.2015.03.015. Biophys J. 2015. PMID: 25902445 Free PMC article.
-
Escape band in Escherichia coli chemotaxis in opposing attractant and nutrient gradients.Proc Natl Acad Sci U S A. 2019 Feb 5;116(6):2253-2258. doi: 10.1073/pnas.1808200116. Epub 2019 Jan 23. Proc Natl Acad Sci U S A. 2019. PMID: 30674662 Free PMC article.
-
Direct measurement of dynamic attractant gradients reveals breakdown of the Patlak-Keller-Segel chemotaxis model.Proc Natl Acad Sci U S A. 2024 Jan 16;121(3):e2309251121. doi: 10.1073/pnas.2309251121. Epub 2024 Jan 9. Proc Natl Acad Sci U S A. 2024. PMID: 38194458 Free PMC article.
References
-
- Adler J, Tso WW (1974) ‘Decision'-making in bacteria: chemotactic response of Escherichia coli to conflicting stimuli. Science 184: 1292–1294 - PubMed
-
- Anraku Y (1968) Transport of sugars and amino acids in bacteria. I. Purification and specificity of the galactose- and leucine-binding proteins. J Biol Chem 243: 3116–3122 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

