Role of a cdk5-associated protein, p35, in herpes simplex virus type 1 replication in vivo
- PMID: 20839922
- PMCID: PMC2975483
- DOI: 10.3109/13550284.2010.513030
Role of a cdk5-associated protein, p35, in herpes simplex virus type 1 replication in vivo
Abstract
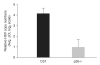
Previous studies have shown that herpes simplex virus type 1 (HSV-1) replication is inhibited by the cyclin-dependent kinase (cdk) inhibitor roscovitine. One roscovitine-sensitive cdk that functions in neurons is cdk5, which is activated in part by its binding partner, p35. Because HSV establishes latent infections in sensory neurons, we sought to determine the role p35 plays in HSV-1 replication in vivo. For these studies, wild-type (wt) and p35−/− mice were infected with HSV-1 using the mouse ocular model of HSV latency and reactivation. The current results indicate that p35 is an important determinant of viral replication in vivo.
Figures



Similar articles
-
Herpes Simplex Virus 1 MicroRNA miR-H8 Is Dispensable for Latency and Reactivation In Vivo.J Virol. 2021 Jan 28;95(4):e02179-20. doi: 10.1128/JVI.02179-20. Print 2021 Jan 28. J Virol. 2021. PMID: 33208453 Free PMC article.
-
An M2 Rather than a TH2 Response Contributes to Better Protection against Latency Reactivation following Ocular Infection of Naive Mice with a Recombinant Herpes Simplex Virus 1 Expressing Murine Interleukin-4.J Virol. 2018 Apr 27;92(10):e00051-18. doi: 10.1128/JVI.00051-18. Print 2018 May 15. J Virol. 2018. PMID: 29491152 Free PMC article.
-
The Absence of DHHC3 Affects Primary and Latent Herpes Simplex Virus 1 Infection.J Virol. 2018 Jan 30;92(4):e01599-17. doi: 10.1128/JVI.01599-17. Print 2018 Feb 15. J Virol. 2018. PMID: 29187538 Free PMC article.
-
[Mechanisms of herpes simplex virus latency and reactivation].Zhejiang Da Xue Xue Bao Yi Xue Ban. 2019 May 25;48(1):89-101. doi: 10.3785/j.issn.1008-9292.2019.02.14. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2019. PMID: 31102363 Free PMC article. Review. Chinese.
-
Immunity in latent Herpes simplex virus infection.Acta Virol. 2005;49(3):159-67. Acta Virol. 2005. PMID: 16178513 Review.
Cited by
-
Herpes simplex virus 1 upregulates p35, alters CDK-5 localization, and stimulates CDK-5 kinase activity during acute infection in neurons.J Virol. 2015 May;89(9):5171-5. doi: 10.1128/JVI.00106-15. Epub 2015 Feb 18. J Virol. 2015. PMID: 25694605 Free PMC article.
-
Phosphorylated-tau associates with HSV-1 chromatin and correlates with nuclear speckles decondensation in low-density host chromatin regions.Neurobiol Dis. 2025 Mar;206:106804. doi: 10.1016/j.nbd.2025.106804. Epub 2025 Jan 14. Neurobiol Dis. 2025. PMID: 39818277 Free PMC article.
-
Rabbit and mouse models of HSV-1 latency, reactivation, and recurrent eye diseases.J Biomed Biotechnol. 2012;2012:612316. doi: 10.1155/2012/612316. Epub 2012 Oct 2. J Biomed Biotechnol. 2012. PMID: 23091352 Free PMC article. Review.
-
Animal Models in Eye Research: Focus on Corneal Pathologies.Int J Mol Sci. 2023 Nov 23;24(23):16661. doi: 10.3390/ijms242316661. Int J Mol Sci. 2023. PMID: 38068983 Free PMC article. Review.
-
Inhibition of Cyclin-dependent Kinase 2 Signaling Prevents Liver Ischemia and Reperfusion Injury.Transplantation. 2019 Apr;103(4):724-732. doi: 10.1097/TP.0000000000002614. Transplantation. 2019. PMID: 30801519 Free PMC article.
References
-
- Chae T, Kwon YT, Bronson R, Dikkes P, Li E, Tsai LH. Mice lacking p35, a neuronal specific activator of Cdk5, display cortical lamination defects, seizures, and adult lethality. Neuron. 1997;18:29–42. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

