Knockdown and overexpression of Unc-45b result in defective myofibril organization in skeletal muscles of zebrafish embryos
- PMID: 20849610
- PMCID: PMC2954953
- DOI: 10.1186/1471-2121-11-70
Knockdown and overexpression of Unc-45b result in defective myofibril organization in skeletal muscles of zebrafish embryos
Abstract
Background: Unc-45 is a myosin chaperone and a Hsp90 co-chaperone that plays a key role in muscle development. Genetic and biochemical studies in C. elegans have demonstrated that Unc-45 facilitates the process of myosin folding and assembly in body wall muscles. Loss or overexpression of Unc-45 in C. elegans results in defective myofibril organization. In the zebrafish Danio rerio, unc-45b, a homolog of C. elegans unc-45, is expressed in both skeletal and cardiac muscles. Earlier studies indicate that mutation or knockdown of unc-45b expression in zebrafish results in a phenotype characterized by a loss of both thick and thin filament organization in skeletal and cardiac muscle. The effects of unc-45b knockdown on other sarcomeric structures and the phenotype of Unc-45b overexpression, however, are poorly understood in vertebrates.
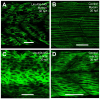
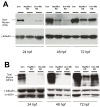
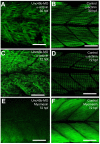
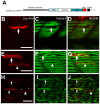
Results: Both knockdown and overexpression provide useful tools to study gene function during animal development. Using such methods, we characterized the role of Unc-45b in myofibril assembly of skeletal muscle in Danio rerio. We showed that, in addition to thick and thin filament defects, knockdown of unc-45b expression disrupted sarcomere organization in M-lines and Z-lines of skeletal muscles in zebrafish embryos. Western blotting analysis showed that myosin protein levels were significantly decreased in unc-45b knockdown embryos. Similarly, embryos overexpressing Unc-45b also exhibited severely disorganized myosin thick filaments. Disruption of thick filament organization by Unc-45b overexpression depends on the C-terminal UCS domain in Unc-45b required for interaction with myosin. Deletion of the C-terminal UCS domain abolished the disruptive activity of Unc-45b in myosin thick filament organization. In contrast, deletion of the N-terminal TPR domain required for binding with Hsp90α had no effect.
Conclusion: Collectively, these studies indicate that the expression levels of Unc-45b must be precisely regulated to ensure normal myofibril organization. Loss or overexpression of Unc-45b leads to defective myofibril organization.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

