Assessment of dexrazoxane as a cardioprotectant in doxorubicin-treated children with high-risk acute lymphoblastic leukaemia: long-term follow-up of a prospective, randomised, multicentre trial
- PMID: 20850381
- PMCID: PMC3756093
- DOI: 10.1016/S1470-2045(10)70204-7
Assessment of dexrazoxane as a cardioprotectant in doxorubicin-treated children with high-risk acute lymphoblastic leukaemia: long-term follow-up of a prospective, randomised, multicentre trial
Abstract
Background: Doxorubicin chemotherapy is associated with cardiomyopathy. Dexrazoxane reduces cardiac damage during treatment with doxorubicin in children with acute lymphoblastic leukaemia (ALL). We aimed to establish the long-term effect of dexrazoxane on the subclinical state of cardiac health in survivors of childhood high-risk ALL 5 years after completion of doxorubicin treatment.
Methods: Between January, 1996, and September, 2000, children with high-risk ALL were enrolled from nine centres in the USA, Canada, and Puerto Rico. Patients were assigned by block randomisation to receive ten doses of 30 mg/m² doxorubicin alone or the same dose of doxorubicin preceded by 300 mg/m² dexrazoxane. Treatment assignment was obtained through a telephone call to a centralised registrar to conceal allocation. Investigators were masked to treatment assignment but treating physicians and patients were not; however, investigators, physicians, and patients were masked to study serum cardiac troponin-T concentrations and echocardiographic measurements. The primary endpoints were late left ventricular structure and function abnormalities as assessed by echocardiography; analyses were done including all patients with data available after treatment completion. This trial has been completed and is registered with ClinicalTrials.gov, number NCT00165087.
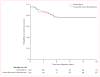
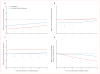
Findings: 100 children were assigned to doxorubicin (66 analysed) and 105 to doxorubicin plus dexrazoxane (68 analysed). 5 years after the completion of doxorubicin chemotherapy, mean left ventricular fractional shortening and end-systolic dimension Z scores were significantly worse than normal for children who received doxorubicin alone (left ventricular fractional shortening: -0·82, 95% CI -1·31 to -0·33; end-systolic dimension: 0·57, 0·21-0·93) but not for those who also received dexrazoxane (-0·41, -0·88 to 0·06; 0·15, -0·20 to 0·51). The protective effect of dexrazoxane, relative to doxorubicin alone, on left ventricular wall thickness (difference between groups: 0·47, 0·46-0·48) and thickness-to-dimension ratio (0·66, 0·64-0·68) were the only statistically significant characteristics at 5 years. Subgroup analysis showed dexrazoxane protection (p=0·04) for left ventricular fractional shortening at 5 years in girls (1·17, 0·24-2·11), but not in boys (-0·10, -0·87 to 0·68). Similarly, subgroup analysis showed dexrazoxane protection (p=0·046) for the left ventricular thickness-to-dimension ratio at 5 years in girls (1·15, 0·44-1·85), but not in boys (0·19, -0·42 to 0·81). With a median follow-up for recurrence and death of 8·7 years (range 1·3-12·1), event-free survival was 77% (95% CI 67-84) for children in the doxorubicin-alone group, and 76% (67-84) for children in the doxorubicin plus dexrazoxane group (p=0·99).
Interpretation: Dexrazoxane provides long-term cardioprotection without compromising oncological efficacy in doxorubicin-treated children with high-risk ALL. Dexrazoxane exerts greater long-term cardioprotective effects in girls than in boys.
Funding: US National Institutes of Health, Children's Cardiomyopathy Foundation, University of Miami Women's Cancer Association, Lance Armstrong Foundation, Roche Diagnostics, Pfizer, and Novartis.
Copyright © 2010 Elsevier Ltd. All rights reserved.
Conflict of interest statement
SEL has received investigator-initiated grants from Pfizer, Novartis, and Roche Diagnostics to help support this study. Pfizer manufactures both doxorubicin and dexrazoxane. Novartis manufactures dexrazoxane in parts of the world outside the USA. Roche Diagnostics manufactures the immunoassay and analyser used to measure cardiac troponin T in the patient’s serum. LBS has received payment for consultancy work for Enzon Pharmaceuticals and ELISA Pharmaceuticals. EVB has received payment for consultancy work, and has stock or stock options, for Genzyme Corporation. All other authors declared that they have no conflicts of interest.
Figures

References
-
- Silverman LB, Gelber RD, Dalton VK, et al. Improved outcome for children with acute lymphoblastic leukemia: results of Dana–Farber Consortium Protocol 91–01. Blood. 2001;97:1211–18. - PubMed
-
- Schrappe M, Reiter A, Ludwig WD, et al. Improved outcome in childhood acute lymphoblastic leukemia despite reduced use of anthracyclines and cranial radiotherapy: results of trial ALL-BFM 90. German-Austrian-Swiss ALL-BFM Study Group. Blood. 2000;95:3310–22. - PubMed
-
- Gaynon PS, Trigg ME, Heerema NA, et al. Children’s Cancer Group trials in childhood acute lymphoblastic leukemia: 1983–1995. Leukemia. 2000;14:2223–33. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- U01 HD052104/HD/NICHD NIH HHS/United States
- HL078522/HL/NHLBI NIH HHS/United States
- HL095127/HL/NHLBI NIH HHS/United States
- R01 HL087000/HL/NHLBI NIH HHS/United States
- HL094100/HL/NHLBI NIH HHS/United States
- F31 HL094100/HL/NHLBI NIH HHS/United States
- HD052102/HD/NICHD NIH HHS/United States
- HL053392/HL/NHLBI NIH HHS/United States
- HL087000/HL/NHLBI NIH HHS/United States
- R01 CA127642/CA/NCI NIH HHS/United States
- HL079233/HL/NHLBI NIH HHS/United States
- HL007188/HL/NHLBI NIH HHS/United States
- R01 HL095127/HL/NHLBI NIH HHS/United States
- K30 HL004537/HL/NHLBI NIH HHS/United States
- T32 HL007188/HL/NHLBI NIH HHS/United States
- HD80002/HD/NICHD NIH HHS/United States
- HL087708/HL/NHLBI NIH HHS/United States
- CA068484/CA/NCI NIH HHS/United States
- R01 HL078522/HL/NHLBI NIH HHS/United States
- AI50274/AI/NIAID NIH HHS/United States
- U01 HD052102/HD/NICHD NIH HHS/United States
- R13 HL087708/HL/NHLBI NIH HHS/United States
- U01 AI050274/AI/NIAID NIH HHS/United States
- HD052104/HD/NICHD NIH HHS/United States
- CA127642/CA/NCI NIH HHS/United States
- P01 CA068484/CA/NCI NIH HHS/United States
- HL072705/HL/NHLBI NIH HHS/United States
- R01 HL053392/HL/NHLBI NIH HHS/United States
- HL004537/HL/NHLBI NIH HHS/United States
- R01 HL072705/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

