Dysregulation of p38 and MKP-1 in response to NOD1/TLR4 stimulation in sarcoid bronchoalveolar cells
- PMID: 20851927
- PMCID: PMC5450927
- DOI: 10.1164/rccm.201005-0792OC
Dysregulation of p38 and MKP-1 in response to NOD1/TLR4 stimulation in sarcoid bronchoalveolar cells
Abstract
Rationale: Sarcoidosis is a systemic inflammatory disorder characterized by distinct up-regulation of Th1 cytokines, such as tumor necrosis factor (TNF)-α and IL-12. The mechanism underlying this up-regulation remains unclear. Recognition of microbial moieties through Toll-like or Nod-like receptors evokes sequential activation of mitogen-activated protein kinases (MAPKs), which plays a role in Th1-immune response.
Objectives: To test the hypothesis that dysregulation in MAPK signaling in response to microbial stimulation is important in mediating Th1 response in sarcoidosis.
Methods: Ex vivo cultured bronchoalveolar lavage (BAL) cells isolated from patients with sarcoidosis and control subjects were stimulated with low-dose Toll-like receptor 4 (TLR4) and nucleotide-binding oligomerization domain 1 (NOD1) ligands as a model of microbial stimulation, and MAPK signaling and inflammatory response were analyzed.
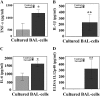
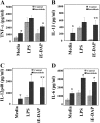
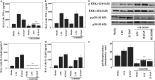
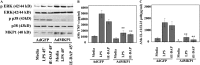
Measurements and main results: BAL cells from patients with sarcoidosis exhibited higher basal p38 activity, greater p38 phosphorylation, and more robust production of TNF-α and IL-12/IL-23p40 on stimulation with NOD1 and TLR4 agonists than cells isolated from control subjects. In contrast, control BAL cells had greater basal extracellular signal-regulated kinase (ERK) activity and NOD1 and TLR4 agonists preferentially activated the ERK pathway. Inhibition of p38, but not ERK, attenuated production of both IL12/IL23p40 and TNF-α. Interestingly, stimulation of cells from patients with sarcoidosis with either NOD1 or TLR4 ligand failed to induce MAPK phosphatase 1 (MKP-1). Adenovirus-mediated overexpression of MKP-1 attenuated p38 activation and decreased the production of IL12/IL23p40 and TNF-α in sarcoid BAL cells.
Conclusions: Our results suggest that enhanced p38 signaling in response to microbial products is caused by abnormal regulation of MKP-1 and contributes to heightened inflammation in sarcoidosis.
Figures


Comment in
-
Innate pathways shape sarcoidosis signaling: from bugs to drugs.Am J Respir Crit Care Med. 2011 Feb 15;183(4):425-7. doi: 10.1164/rccm.201010-1570ED. Am J Respir Crit Care Med. 2011. PMID: 21325080 No abstract available.
Similar articles
-
Mitogen-activated protein kinase phosphatase (MKP)-1 in immunology, physiology, and disease.Life Sci. 2012 Feb 13;90(7-8):237-48. doi: 10.1016/j.lfs.2011.11.017. Epub 2011 Dec 13. Life Sci. 2012. PMID: 22197448 Free PMC article. Review.
-
Early p38 Activation Regulated by MKP-1 Is Determinant for High Levels of IL-10 Expression Through TLR2 Activation.Front Immunol. 2021 Jun 21;12:660065. doi: 10.3389/fimmu.2021.660065. eCollection 2021. Front Immunol. 2021. PMID: 34234775 Free PMC article.
-
The Role of the p38-MNK-eIF4E Signaling Axis in TNF Production Downstream of the NOD1 Receptor.J Immunol. 2017 Feb 15;198(4):1638-1648. doi: 10.4049/jimmunol.1600467. Epub 2017 Jan 13. J Immunol. 2017. PMID: 28087669
-
Functional Toll-Like Receptor 9 Expression and CXCR3 Ligand Release in Pulmonary Sarcoidosis.Am J Respir Cell Mol Biol. 2016 Nov;55(5):749-757. doi: 10.1165/rcmb.2015-0278OC. Am J Respir Cell Mol Biol. 2016. PMID: 27390897
-
Acetylation of MKP-1 and the control of inflammation.Sci Signal. 2008 Oct 14;1(41):pe44. doi: 10.1126/scisignal.141pe44. Sci Signal. 2008. PMID: 18922786 Free PMC article. Review.
Cited by
-
Metabolomics connects aberrant bioenergetic, transmethylation, and gut microbiota in sarcoidosis.Metabolomics. 2016 Feb;12:35. doi: 10.1007/s11306-015-0932-2. Epub 2016 Jan 20. Metabolomics. 2016. PMID: 27489531 Free PMC article.
-
NOD1 and NOD2 in inflammatory and infectious diseases.Immunol Rev. 2020 Sep;297(1):139-161. doi: 10.1111/imr.12902. Epub 2020 Jul 17. Immunol Rev. 2020. PMID: 32677123 Free PMC article. Review.
-
Modulatory role of macrophage migration inhibitory factor on cytokines and clinical features of sarcoidosis.Sci Rep. 2022 Oct 7;12(1):16906. doi: 10.1038/s41598-022-21212-5. Sci Rep. 2022. PMID: 36207373 Free PMC article.
-
Mitogen-activated protein kinase phosphatase (MKP)-1 in immunology, physiology, and disease.Life Sci. 2012 Feb 13;90(7-8):237-48. doi: 10.1016/j.lfs.2011.11.017. Epub 2011 Dec 13. Life Sci. 2012. PMID: 22197448 Free PMC article. Review.
-
Lung fibrosis in sarcoidosis. Is there a place for antifibrotics?Front Pharmacol. 2024 Aug 30;15:1445923. doi: 10.3389/fphar.2024.1445923. eCollection 2024. Front Pharmacol. 2024. PMID: 39281278 Free PMC article. Review.
References
-
- Statement on sarcoidosis. Joint statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999. Am J Respir Crit Care Med 1999;160:736–755. - PubMed
-
- Prior C, Knight RA, Herold M, Ott G, Spiteri MA. Pulmonary sarcoidosis: patterns of cytokine release in vitro. Eur Respir J 1996;9:47–53. - PubMed
-
- Furukawa A, Uchida K, Ishige Y, Ishige I, Kobayashi I, Takemura T, Yokoyama T, Iwai K, Watanabe K, Shimizu S, et al. Characterization of propionibacterium acnes isolates from sarcoid and non-sarcoid tissues with special reference to cell invasiveness, serotype, and trigger factor gene polymorphism. Microb Pathog 2009;46:80–87. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

