Chylomicron- and VLDL-derived lipids enter the heart through different pathways: in vivo evidence for receptor- and non-receptor-mediated fatty acid uptake
- PMID: 20852327
- PMCID: PMC2992231
- DOI: 10.1074/jbc.M110.174458
Chylomicron- and VLDL-derived lipids enter the heart through different pathways: in vivo evidence for receptor- and non-receptor-mediated fatty acid uptake
Abstract
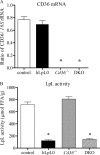
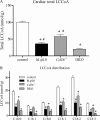
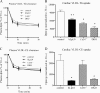
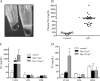
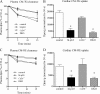
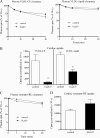
Lipids circulate in the blood in association with plasma lipoproteins and enter the tissues either after hydrolysis or as non-hydrolyzable lipid esters. We studied cardiac lipids, lipoprotein lipid uptake, and gene expression in heart-specific lipoprotein lipase (LpL) knock-out (hLpL0), CD36 knock-out (Cd36(-/-)), and double knock-out (hLpL0/Cd36(-/-)-DKO) mice. Loss of either LpL or CD36 led to a significant reduction in heart total fatty acyl-CoA (control, 99.5 ± 3.8; hLpL0, 36.2 ± 3.5; Cd36(-/-), 57.7 ± 5.5 nmol/g, p < 0.05) and an additive effect was observed in the DKO (20.2 ± 1.4 nmol/g, p < 0.05). Myocardial VLDL-triglyceride (TG) uptake was reduced in the hLpL0 (31 ± 6%) and Cd36(-/-) (47 ± 4%) mice with an additive reduction in the DKO (64 ± 5%) compared with control. However, LpL but not CD36 deficiency decreased VLDL-cholesteryl ester uptake. Endogenously labeled mouse chylomicrons were produced by tamoxifen treatment of β-actin-MerCreMer/LpL(flox/flox) mice. Induced loss of LpL increased TG levels >10-fold and reduced HDL by >50%. After injection of these labeled chylomicrons in the different mice, chylomicron TG uptake was reduced by ∼70% and retinyl ester by ∼50% in hLpL0 hearts. Loss of CD36 did not alter either chylomicron TG or retinyl ester uptake. LpL loss did not affect uptake of remnant lipoproteins from ApoE knock-out mice. Our data are consistent with two pathways for fatty acid uptake; a CD36 process for VLDL-derived fatty acid and a non-CD36 process for chylomicron-derived fatty acid uptake. In addition, our data show that lipolysis is involved in uptake of core lipids from TG-rich lipoproteins.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
- HL73029/HL/NHLBI NIH HHS/United States
- R01 DK079221/DK/NIDDK NIH HHS/United States
- R01 HL045095/HL/NHLBI NIH HHS/United States
- DK079221/DK/NIDDK NIH HHS/United States
- HHMI/Howard Hughes Medical Institute/United States
- P30 DK056341/DK/NIDDK NIH HHS/United States
- U24 DK059635/DK/NIDDK NIH HHS/United States
- R01 DK040936/DK/NIDDK NIH HHS/United States
- AA019413/AA/NIAAA NIH HHS/United States
- RC2 AA019413/AA/NIAAA NIH HHS/United States
- R37 HL045095/HL/NHLBI NIH HHS/United States
- R01 DK033301/DK/NIDDK NIH HHS/United States
- HL45095/HL/NHLBI NIH HHS/United States
- P01 HL057278/HL/NHLBI NIH HHS/United States
- R01 HL073029/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

