Physiology and Endocrinology of the Ovarian Cycle in Macaques
- PMID: 20852722
- PMCID: PMC2939751
- DOI: 10.1177/0192623308327412
Physiology and Endocrinology of the Ovarian Cycle in Macaques
Abstract
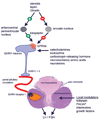
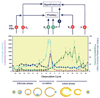
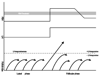
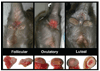
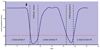
Macaques provide excellent models for preclinical testing and safety assessment of female reproductive toxicants. Currently, cynomolgus monkeys are the predominant species for (reproductive) toxicity testing. Marmosets and rhesus monkeys are being used occasionally. The authors provide a brief review on physiology and endocrinology of the cynomolgus monkey ovarian cycle, practical guidance on assessment and monitoring of ovarian cyclicity, and new data on effects of social housing on ovarian cyclicity in toxicological studies. In macaques, cycle monitoring is achieved using daily vaginal smears for menstruation combined with cycle-timed frequent sampling for steroid and peptide hormone analysis. Owing to requirements of frequent and timed blood sampling, it is not recommended to incorporate these special evaluations into a general toxicity study design. Marmosets lack external signs of ovarian cyclicity, and cycle monitoring is done by regular determinations of progesterone. Cynomolgus and marmoset monkeys do not exhibit seasonal variations in ovarian activity, whereas such annual rhythm is pronounced in rhesus monkeys. Studies on pair- and group-housed cynomolgus monkeys revealed transient alterations in the duration and endocrinology of the ovarian cycle followed by return to normal cyclicity after approximately six months. This effect is avoided if the animals had contact with each other prior to mingling. These experiments also demonstrated that synchronization of ovarian cycles did not occur.
Figures



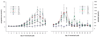




References
-
- Abbott DH, Barnett DK, Colman RJ, Yamamoto ME, Schultz-Darken NJ. Aspects of common marmoset basic biology and life history important for biomedical research. Comp Med. 2003;53:339–350. - PubMed
-
- Abbott DH, Saltzman W, Schultz-Darken NJ, Smith TE. Specific neuroendocrine mechanisms not involving generalized stress mediate social regulation of female reproduction in cooperatively breeding marmoset monkeys. Ann NY Acad Sci. 1997;807:219–238. - PubMed
-
- Adams MR, Kaplan JR, Koritnik DR. Psychosocial influences on ovarian endocrine and ovulatory function in Macaca fascicularis. Physiol Behav. 1985;35:935–940. - PubMed
-
- Archer DF. Role of the nonhuman primate for research related to women’s health. Ilar J. 2004;45:212–219. - PubMed
-
- Attia MA. Cyclic changes in genital organs and vaginal cytology in cynomolgus monkeys (Macaca fascicularis) Dtsch Tierarztl Wochenschr. 1998;105:399–404. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

