Role of ubiquitination in endocytic trafficking of G-protein-coupled receptors
- PMID: 20854416
- PMCID: PMC3625703
- DOI: 10.1111/j.1600-0854.2010.01121.x
Role of ubiquitination in endocytic trafficking of G-protein-coupled receptors
Abstract
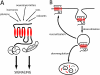
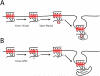
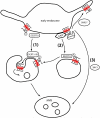
Lysyl ubiquitination has long been known to target cytoplasmic proteins for proteasomal degradation, and there is now extensive evidence that ubiquitination functions in vacuolar/lysosomal targeting of membrane proteins from both the biosynthetic and endocytic pathways. G-protein-coupled receptors (GPCRs) represent the largest and most diverse family of membrane proteins, whose function is of fundamental importance both physiologically and therapeutically. In this review, we discuss the role of ubiquitination in the vacuolar/lysosomal downregulation of GPCRs through the endocytic pathway, with a primary focus on lysosomal trafficking in mammalian cells. We will summarize evidence indicating that mammalian GPCRs are regulated by ubiquitin-dependent mechanisms conserved in budding yeast, and then consider evidence for additional ubiquitin-dependent and -independent regulation that may be specific to animal cells.
© 2010 John Wiley & Sons A/S.
Figures
References
-
- Jacoby E, Bouhelal R, Gerspacher M, Seuwen K. The 7 TM G-protein-coupled receptor target family. ChemMedChem. 2006;1(8):761–782. - PubMed
-
- Fredholm BB, Hokfelt T, Milligan G. G-protein-coupled receptors: an update. Acta Physiol (Oxf) 2007;190(1):3–7. - PubMed
-
- Brzostowski JA, Kimmel AR. Signaling at zero G: G-protein-independent functions for 7-TM receptors. Trends Biochem Sci. 2001;26(5):291–297. - PubMed
-
- DeWire SM, Ahn S, Lefkowitz RJ, Shenoy SK. Beta-arrestins and cell signaling. Annu Rev Physiol. 2007;69:483–510. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

