Endothelial IL-1R1 is a critical mediator of EAE pathogenesis
- PMID: 20854891
- PMCID: PMC2991628
- DOI: 10.1016/j.bbi.2010.09.009
Endothelial IL-1R1 is a critical mediator of EAE pathogenesis
Abstract
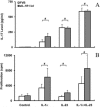
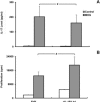
Interleukin-1 (IL-1) has been implicated in the disease progression of multiple sclerosis (MS). In the animal model of MS, experimental autoimmune encephalomyelitis (EAE), the induction of disease is significantly attenuated in mice lacking the type I IL-1 receptor (IL-1R1). In this study, we created a transgenic mouse (eIL-1R1 kd) in which IL-1R1 expression is knocked down specifically in endothelial cells. Induction of EAE in eIL-1R1 kd mice results in a decrease in incidence, severity and delayed onset of EAE. In addition, eIL-1R1 kd mice show significant decrease in VCAM-1 expression and diminished CD45(+) and CD3(+) infiltrating leukocytes in the spinal cord in animals challenged with EAE. Further, IL-1 and IL-23 stimulate IL-17 production by splenocytes from both wild type and the eIL-1R1 kd animals. Similarly, IL-1 and IL-23 synergistically stimulate splenocytes proliferation in these two strains of animals. After immunization with MOG(79-96), although eIL-1R1 kd mice displayed greatly reduced clinical scores, their splenocytes produced IL-17 and proliferated in response to a second MOG challenge, similar to wild type animals. These findings indicate a critical role for endothelial IL-1R1 in mediating the pathogenesis of EAE, and describe a new model that can be used to study endothelial IL-1R1.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures

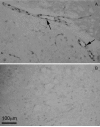
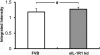
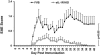
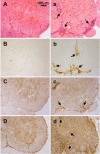
Similar articles
-
Superantigen overcomes resistance of IL-6-deficient mice towards MOG-induced EAE by a TNFR1 controlled pathway.Eur J Immunol. 2001 Aug;31(8):2302-12. doi: 10.1002/1521-4141(200108)31:8<2302::aid-immu2302>3.0.co;2-#. Eur J Immunol. 2001. PMID: 11477542
-
IL-6-deficient mice resist myelin oligodendrocyte glycoprotein-induced autoimmune encephalomyelitis.Eur J Immunol. 1998 Jul;28(7):2178-87. doi: 10.1002/(SICI)1521-4141(199807)28:07<2178::AID-IMMU2178>3.0.CO;2-D. Eur J Immunol. 1998. PMID: 9692887
-
Liver X receptor agonist regulation of Th17 lymphocyte function in autoimmunity.J Leukoc Biol. 2009 Aug;86(2):401-9. doi: 10.1189/jlb.1008600. J Leukoc Biol. 2009. PMID: 19406833 Free PMC article.
-
Actin-Binding Protein Cortactin Promotes Pathogenesis of Experimental Autoimmune Encephalomyelitis by Supporting Leukocyte Infiltration into the Central Nervous System.J Neurosci. 2020 Feb 12;40(7):1389-1404. doi: 10.1523/JNEUROSCI.1266-19.2019. Epub 2020 Jan 7. J Neurosci. 2020. PMID: 31911458 Free PMC article.
-
Myelin oligodendrocyte glycoprotein (MOG35-55)-induced experimental autoimmune encephalomyelitis is ameliorated in interleukin-32 alpha transgenic mice.Oncotarget. 2015 Dec 1;6(38):40452-63. doi: 10.18632/oncotarget.6306. Oncotarget. 2015. PMID: 26564962 Free PMC article.
Cited by
-
Interleukin-1 promotes autoimmune neuroinflammation by suppressing endothelial heme oxygenase-1 at the blood-brain barrier.Acta Neuropathol. 2020 Oct;140(4):549-567. doi: 10.1007/s00401-020-02187-x. Epub 2020 Jul 11. Acta Neuropathol. 2020. PMID: 32651669 Free PMC article.
-
New Insights into the Role of IL-1β in Experimental Autoimmune Encephalomyelitis and Multiple Sclerosis.J Immunol. 2017 Jun 15;198(12):4553-4560. doi: 10.4049/jimmunol.1700263. J Immunol. 2017. PMID: 28583987 Free PMC article. Review.
-
Hypoxia-like tissue injury and glial response contribute to Balo concentric lesion development.Neurology. 2016 Nov 8;87(19):2000-2005. doi: 10.1212/WNL.0000000000003308. Epub 2016 Oct 12. Neurology. 2016. PMID: 27733565 Free PMC article.
-
Schizophrenia in the genetic era: a review from development history, clinical features and genomic research approaches to insights of susceptibility genes.Metab Brain Dis. 2024 Jan;39(1):147-171. doi: 10.1007/s11011-023-01271-x. Epub 2023 Aug 5. Metab Brain Dis. 2024. PMID: 37542622 Review.
-
Astrocytic YAP protects the optic nerve and retina in an experimental autoimmune encephalomyelitis model through TGF-β signaling.Theranostics. 2021 Jul 25;11(17):8480-8499. doi: 10.7150/thno.60031. eCollection 2021. Theranostics. 2021. PMID: 34373754 Free PMC article.
References
-
- Ahmed ST, Mayer A, Ji JD, Ivashkiv LB. Inhibition of IL-6 signaling by a p38-dependent pathway occurs in the absence of new protein synthesis. J Leukoc Biol. 2002;72(1):154–162. - PubMed
-
- Badovinac V, Mostarica-Stojkovic M, Dinarello CA, Stosic-Grujicic S. Interleukin-1 receptor antagonist suppresses experimental autoimmune encephalomyelitis (EAE) in rats by influencing the activation and proliferation of encephalitogenic cells. J Neuroimmunol. 1998;85(1):87–95. - PubMed
-
- Baker AM, Grekova MC, Richert JR. EAE susceptibility in FVB mice. J Neurosci Res. 2000;61(2):140–145. - PubMed
-
- Ban E, Milon G, Prudhomme N, Fillion G, Haour F. Receptors for interleukin-1 (alpha and beta) in mouse brain: mapping and neuronal localization in hippocampus. Neuroscience. 1991;43(1):21–30. - PubMed
-
- Basu A, Krady JK, Levison SW. Interleukin-1: a master regulator of neuroinflammation. J Neurosci Res. 2004;78(2):151–156. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

